Which compound (s) out of the following is(are) not aromatic?
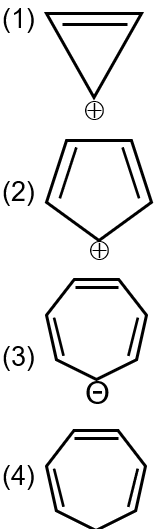
(2)→ antiaromatic
(3) and (4)→ Non aromatic
Aromatic compounds must satisfy Hückel's rule: they must be cyclic, planar, fully conjugated, and contain 4n+2 π electrons (where n is an integer). Non-aromatic compounds fail one or more of these criteria.
Compound (1) is a benzene ring with a methyl group. Benzene is aromatic: it is cyclic, planar, fully conjugated, and has 6 π electrons (n=1, 4*1+2=6). The methyl group does not affect aromaticity. So, (1) is aromatic.
Compound (2) is cyclooctatetraene. It is cyclic and fully conjugated but not planar (it adopts a tub-shaped conformation to avoid angle strain). It has 8 π electrons (n=2, 4*2=8, which does not satisfy 4n+2). Thus, it is non-aromatic.
Compound (3) is cyclopentadienyl anion. It is cyclic, planar, and fully conjugated. It has 6 π electrons (after losing H+, the anion has 6 π electrons, n=1, 4*1+2=6). So, it is aromatic.
Compound (4) is cyclopropenyl cation. It is cyclic, planar, and fully conjugated. It has 2 π electrons (n=0, 4*0+2=2). So, it is aromatic.
Only compound (2) is not aromatic. Therefore, the correct option is (2).
Hückel's Rule: A compound is aromatic if it has π electrons, where is an integer (0, 1, 2, ...).
Conditions for Aromaticity: