Among P, Q, R and S, the aromatic compound(s) is / are
Aromatic compounds follow Hückel's rule: they must be cyclic, planar, fully conjugated, and have 4n+2 π electrons (where n is an integer).
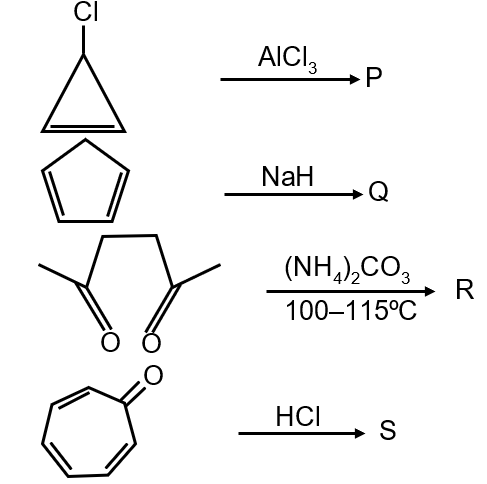
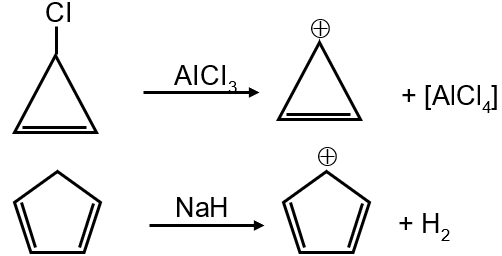
Compound P is a cyclopentadienyl anion. It has 5 carbon atoms with a negative charge. The π electron count: each carbon in the ring contributes one electron, and the negative charge adds two electrons, totaling 6 π electrons. n=1 satisfies 4(1)+2=6. It is cyclic, planar, and conjugated. Thus, P is aromatic.
Compound Q is cycloheptatriene with a positive charge. It has 7 carbon atoms. The positive charge indicates loss of two electrons. The π system has 6 electrons (from three double bonds, but the positive charge reduces count). Actually, it has 6 π electrons (similar to tropylium cation). n=1 satisfies 4(1)+2=6. It is cyclic, planar, and conjugated. Thus, Q is aromatic.
Compound R is cyclooctatetraene. It has 8 π electrons (from four double bonds). For n=2, 4(2)+2=10, which doesn't match. For n=1.5, not integer. It is not aromatic; instead, it is non-aromatic because it adopts a tub conformation to avoid strain, breaking planarity.
Compound S is a cyclopentadienyl cation. It has 5 carbon atoms with a positive charge. The π electron count: only 4 electrons (positive charge removes two). For n=0.5, not integer. It does not satisfy Hückel's rule and is anti-aromatic.
The aromatic compounds are P and Q.
Hückel's Rule: Aromatic if π electrons = , where n is an integer (0,1,2...).
Key properties: Cyclic, planar, fully conjugated π system.
π electron calculation: For charged systems, add or subtract electrons based on charge.