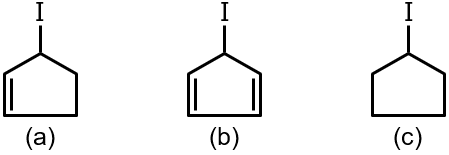
Rate of abstraction of iodine by Ag⊕ is
a > c> b
This question involves comparing the rate of abstraction of iodine by silver ion (Ag⁺) from different alkyl iodides. The rate depends on the stability of the carbocation formed in the SN1 reaction mechanism, as Ag⁺ aids in the dissociation of the C-I bond.
Step 1: Understand the Reaction Mechanism
The reaction is an SN1-type process facilitated by Ag⁺. Ag⁺ coordinates with the iodine atom, helping to break the C-I bond and form a carbocation intermediate. The rate of this reaction is determined by the stability of this carbocation.
General Reaction:
Step 2: Identify the Carbocations Formed
We need to analyze the structure of each reactant (a, b, c) to see the carbocation that would be generated.
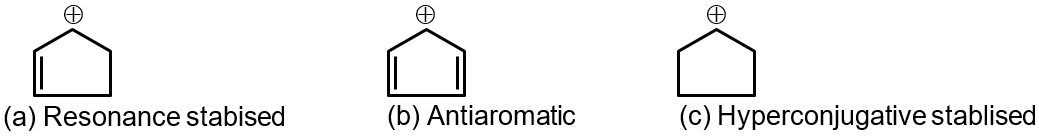
Step 3: Rank the Carbocation Stabilities
The order of carbocation stability is: Tertiary > Secondary > Allylic ≈ Benzyl > Primary > Methyl.
Comparing our cases:
Therefore, the stability order is: a > c > b.
Step 4: Relate Stability to Reaction Rate
A more stable carbocation is formed faster because the transition state leading to it is lower in energy. Therefore, the rate of iodine abstraction follows the same order as carbocation stability.
Final Answer: a > c > b
Related Topics:
Key Formulae and Theory: