Identifying Aromatic Compounds
To determine if a compound is aromatic, we use Hückel's Rule: A compound must be cyclic, planar, fully conjugated, and have 4n+2 π electrons (where n is an integer).
Step 1: Analyze Each Compound
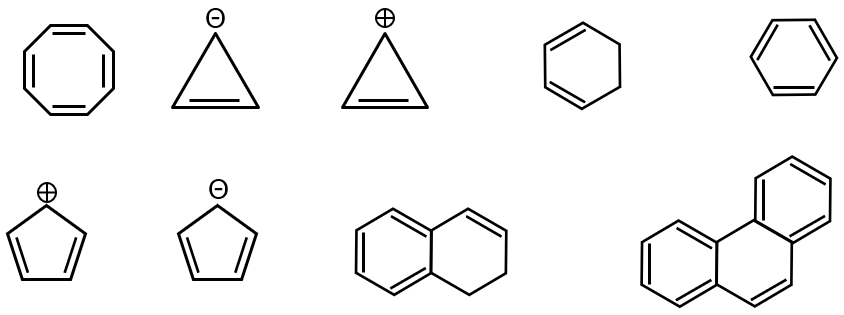
Compound 1: Cyclopentadienyl anion
- Cyclic: Yes
- Planar: Yes
- Fully conjugated: Yes (all carbons are sp² hybridized)
- π electrons: 6 (from 5 carbons contributing one electron each, plus one extra from negative charge). So, 4n+2 with n=1.
Conclusion: Aromatic.
Compound 2: Cycloheptatrienylium cation (Tropylium cation)
- Cyclic: Yes
- Planar: Yes
- Fully conjugated: Yes
- π electrons: 6 (from 6 electrons in triple bonds and positive charge removes one electron, leaving 6). So, 4n+2 with n=1.
Conclusion: Aromatic.
Compound 3: Cyclooctatetraene
- Cyclic: Yes
- Planar: No (adopts a tub conformation to avoid angle strain)
- Fully conjugated: Not fully due to non-planarity
- π electrons: 8 (which is 4n with n=2, not 4n+2).
Conclusion: Not aromatic.
Compound 4: [10]-Annulene
- Cyclic: Yes
- Planar: No (internal hydrogens cause steric hindrance, forcing it out of planarity)
- Fully conjugated: Not fully due to non-planarity
- π electrons: 10 (which is 4n+2 with n=2, but non-planarity disqualifies it).
Conclusion: Not aromatic.
Compound 5: Azulene
- Cyclic: Yes
- Planar: Yes
- Fully conjugated: Yes
- π electrons: 10 (4n+2 with n=2). It has a significant dipole moment and behaves as an aromatic compound.
Conclusion: Aromatic.
Compound 6: Biphenyl (in the context shown, likely the ring system)
- While each benzene ring is aromatic, the entire biphenyl system is not fully conjugated across the single bond (which allows rotation, breaking full conjugation for the entire system). However, each ring individually is aromatic. The question typically considers the entire molecule, which is not aromatic as a whole but contains aromatic rings.
Conclusion: Not aromatic (as a whole molecule).
Step 2: Count the Aromatic Compounds
From the analysis: Compounds 1, 2, and 5 are aromatic. So, there are 3 aromatic compounds.
Final Answer
The number of aromatic compounds is .
Related Topics
- Hückel's Rule
- Antiaromaticity (compounds with 4n π electrons that are unstable)
- Non-aromatic compounds
- Resonance and conjugation
- Molecular orbital theory applied to aromaticity
Formulae and Theory
Hückel's Rule: For a compound to be aromatic, it must have π electrons, where n is an integer (0, 1, 2, ...).
Key Conditions:
- Cyclic
- Planar
- Fully conjugated (every atom in the ring must have a p orbital)
Examples: Benzene (6 π electrons, n=1), Cyclopentadienyl anion (6 π electrons), Tropylium cation (6 π electrons).