The major product in the following reaction is :
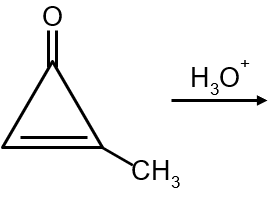
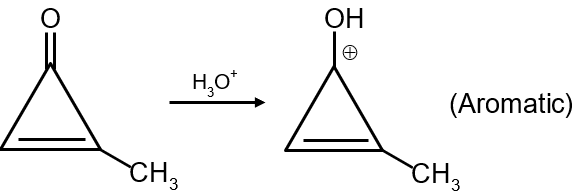
The given reaction shows an alkene undergoing electrophilic addition with HBr. The alkene is unsymmetrical (has different groups attached to each carbon of the double bond), so we must use Markovnikov's rule to predict the major product.
The reactant is 3-methylbut-1-ene. Its structure is: CH2=CH-CH(CH3)2.
Let's label the carbons of the double bond for clarity:
Markovnikov's rule states that in the addition of a protic acid (like HBr) to an unsymmetrical alkene, the hydrogen atom (H+) adds to the carbon of the double bond that has the greater number of hydrogen atoms (i.e., the less substituted carbon).
In our alkene:
Therefore, the H+ from HBr will add to Carbon 1. The Br- ion will then add to the more stable carbocation intermediate that forms on Carbon 2.
After H+ adds to Carbon 1, a carbocation forms on Carbon 2. Let's examine its structure:
This is a secondary carbocation. However, it is stabilized by hyperconjugation from the three alkyl groups attached to it. This makes it a stable intermediate. There is no possibility for a rearrangement to form an even more stable tertiary carbocation in this specific molecule.
The bromide ion (Br-) attacks the carbocation, forming the final alkyl bromide. The major product is 2-bromo-2-methylbutane.
Its structure is: CH3-C(Br)(CH3)-CH3 or (CH3)2C(Br)CH3. This is a tertiary alkyl halide.
The major product is the tertiary bromide: 2-bromo-2-methylbutane.
This corresponds to the third option in the list of possible answers.
Rule: "The rich get richer." In the addition of HX to an unsymmetrical alkene, the hydrogen adds to the carbon that already has more hydrogens.
Mechanism: Electrophilic Addition
General Reaction:
The stability of the carbocation intermediate is the driving force behind Markovnikov's rule. The order of stability is:
Methyl < Primary (1°) < Secondary (2°) < Tertiary (3°) < Resonance stabilized (e.g., allylic, benzylic)
This stability is due to:
In the presence of peroxides (ROOR), HBr adds to alkenes in an anti-Markovnikov fashion. This is due to a free radical mechanism, not an electrophilic one. This exception only applies to HBr; HCl and HI do not show this behavior.