The major product 'X' formed in the following reaction is :
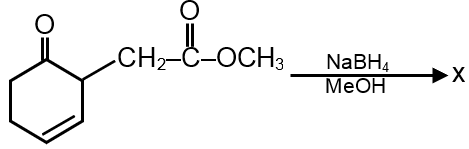
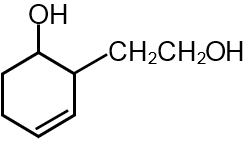
NaBH4 don’t reduce ester.
The given reaction involves a cyclic ketone (cyclopentanone) undergoing a reaction with methyl magnesium bromide (CH3MgBr), followed by hydrolysis. This is a classic example of a Grignard reaction with a ketone.
Step 1: Nucleophilic Addition
Methyl magnesium bromide (a Grignard reagent) acts as a strong nucleophile. The nucleophilic carbon of the CH3- group attacks the electrophilic carbonyl carbon of cyclopentanone. This results in the formation of a magnesium alkoxide intermediate.
Step 2: Hydrolysis
In the subsequent step, the reaction mixture is hydrolyzed (treated with H2O or a dilute acid). This step protonates the alkoxide group, converting it into a hydroxyl group (). The final product is a tertiary alcohol.
Final Answer:
For a ketone, the Grignard reaction produces a tertiary alcohol. Since the starting material is a symmetrical ketone (cyclopentanone), the product is 1-methylcyclopentanol.
Therefore, the correct structure of the major product 'X' is:
Grignard Reagents: Organomagnesium compounds (R-MgX) that are strong nucleophiles and bases. They react with carbonyl compounds to form alcohols.
Reaction Summary:
The general reaction for a ketone with a Grignard reagent is:
Key Point: The reaction is highly moisture-sensitive and must be performed under anhydrous conditions, as Grignard reagents are destroyed by water and protic solvents.