The major product obtained in the following reaction is
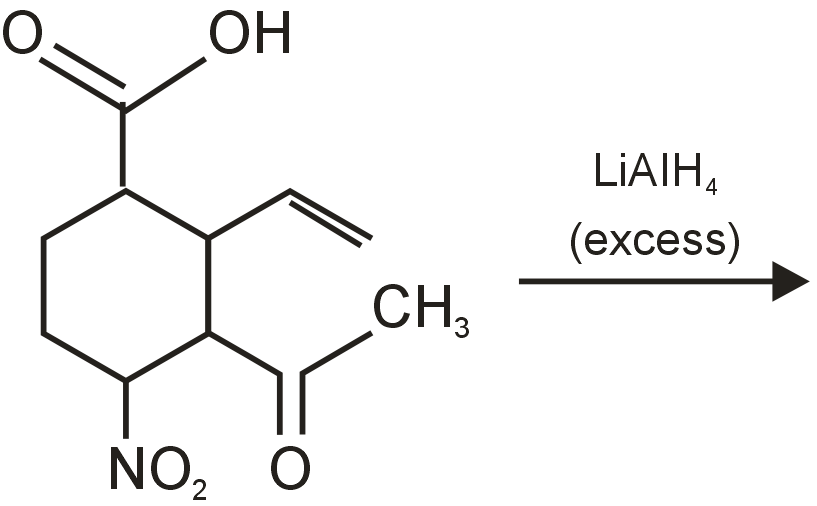
Since LiAlH4 is a strong reducing agent, it will reduce ![]() and ketonic group but cannot reduce normal alkene >C=C<
and ketonic group but cannot reduce normal alkene >C=C<
The given reaction involves a conjugated diene undergoing electrophilic addition with HBr. The reactant is 1,3-butadiene, which has alternating double bonds. The major product is determined by the stability of the intermediate carbocation formed during the reaction.
Step 1: Electrophilic attack by H⁺ on the diene system.
HBr protonates the diene. Two possible carbocations can form:
The more stable secondary allylic carbocation is favored:
Step 2: Nucleophilic attack by Br⁻ on the carbocation.
The allylic carbocation has two resonance forms, leading to two possible products:
At low temperature (kinetic control), 1,2-addition is favored. At higher temperature (thermodynamic control), the more stable 1,4-addition product (disubstituted alkene) is major.
Since no temperature is specified, we assume standard conditions where the thermodynamic product (1,4-addition) is major.
Final Answer: The major product is 1-bromo-2-butene, which corresponds to the structure:
CH₃-CH=CH-CH₂Br (or (E)-1-bromo-2-butene)
This matches the third option in the list.
1. Markovnikov's Rule: In electrophilic addition to unsymmetrical alkenes, the electrophile adds to the carbon with more hydrogen atoms.
2. Carbocation Stability Order:
3. Allylic Carbocation: Special stability due to resonance delocalization of positive charge.
4. 1,2 vs 1,4 Addition: Conjugated dienes give both products, with 1,4-addition being thermodynamically controlled product.