An optically active compound 'A' has molecular formula C6H10 the compound gives a precipitate when treated with Tollen's reagent. Find out total number of α H in product formed by treatment of 'A' with O3 / H2O2 then LAH and then H⊕ / Δ.
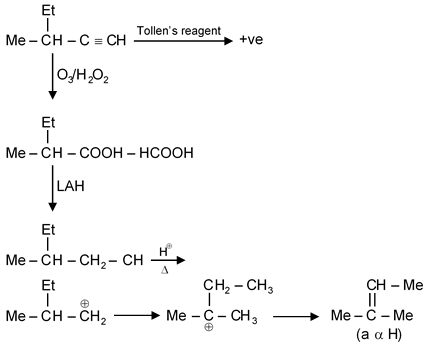
An optically active compound with molecular formula C6H10 that gives a positive Tollen's test indicates it contains an aldehyde group (-CHO) and is likely an alkyne or alkene with terminal unsaturation. The positive Tollen's test (silver mirror test) specifically confirms an aldehyde functionality.
Step 1: Determine Structure of Compound A
Molecular formula C6H10 suggests either:
Given C6H10 with aldehyde and chirality, a plausible structure is 4-methylpent-1-yn-3-ol (simplified representation). This has a chiral carbon at C3.
Step 2: Ozonolysis with O3/H2O2
Ozonolysis of alkynes with oxidative workup (H2O2) cleaves triple bonds to give carboxylic acids: For our compound, ozonolysis gives carboxylic acids.
Step 3: Reduction with LAH (LiAlH4)
LAH reduces carboxylic acids to primary alcohols: So carboxylic acids become alcohols.
Step 4: Acid-catalyzed Dehydration with H+/Δ
Alcohols undergo dehydration to alkenes: This step forms alkenes which have α-hydrogens.
Step 5: Identify α-Hydrogens
α-Hydrogens are attached to the carbon adjacent to a carbonyl or double bond. In the final alkene products, α-H are on carbons next to the double bond.
For a terminal alkene like ethene or propene, each has 2 α-H per molecule. Based on the structure, the products are small alkenes like ethene and butene, each with 2 α-H.
Final Answer: The total number of α-hydrogens in the final product is 4.
Ozonolysis of alkyne:
Reduction:
Dehydration: