|
Column-I (Reaction) |
Column-II (Most suitable Reagent) |
 |
(P) (i) SnCl2/HCl, (ii) H+ / H2O |
 |
(Q) (i) LiAlH4, (ii) H2O |
 |
(R) (CH3)2Cd |
 |
(S) (i) CH3Li (2 eq.), (ii) H2O |
| (T) (i) CH3MgBr (excess), (ii) H2O |
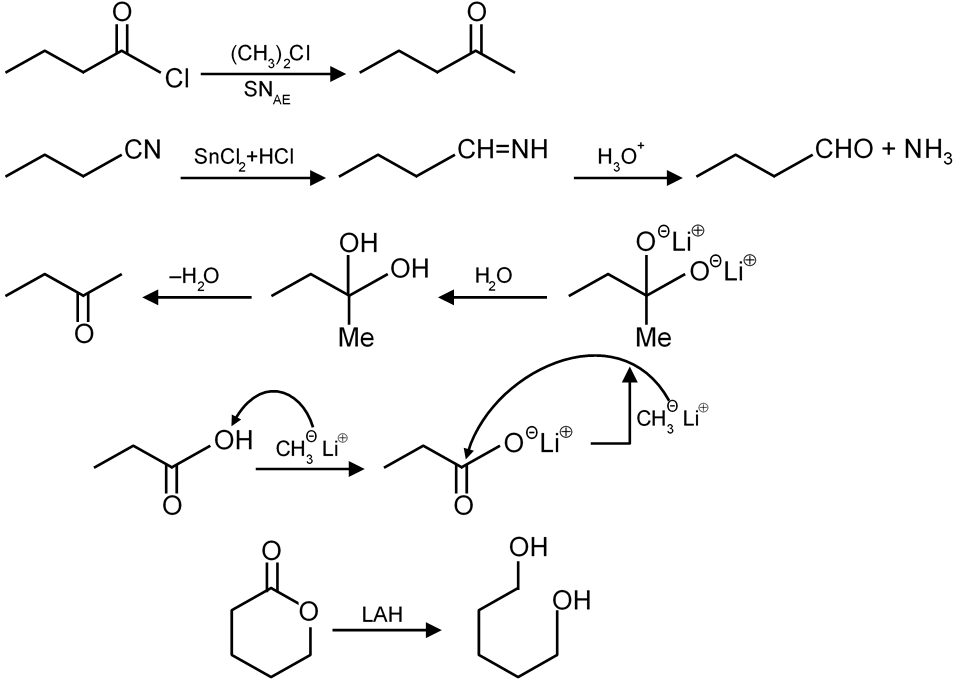
This exercise involves matching specific organic reactions (Column-I) with the most appropriate reagents (Column-II) to achieve the desired transformation. The key is to understand the functional groups involved and the selectivity of each reagent.
Step 1: Analyze the first reaction.
The reactant is a nitro group attached to a benzene ring. The desired product is aniline (NH₂ group).
The best way to reduce a nitro group (NO₂) to an amino group (NH₂) selectively is using Tin(II) Chloride in Hydrochloric acid (SnCl₂/HCl), followed by hydrolysis to get the free amine. This matches with option (P).
Reaction:
Step 2: Analyze the second reaction.
The reactant is a carboxylic acid (COOH). The product is a primary alcohol (CH₂OH).
Lithium Aluminum Hydride (LiAlH₄) is a strong reducing agent that reduces carboxylic acids to primary alcohols. This is followed by hydrolysis. This matches with option (Q).
Reaction:
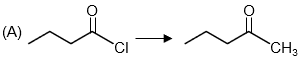
Step 3: Analyze the third reaction.
The reactant is an acid chloride. The product is a ketone where a methyl group has been added.
To convert an acid chloride to a ketone, we need a reagent that acts as a source of a nucleophilic carbon but is not too reactive (to avoid over-addition to a tertiary alcohol). Dialkylcadmium reagents (e.g., (CH₃)₂Cd) are less reactive than Grignard reagents and stop at the ketone stage. This matches with option (R).
Reaction:
Step 4: Analyze the fourth reaction.
The reactant is an ester. The product is a tertiary alcohol where two methyl groups have been added.
Esters react with excess Grignard reagents (like CH₃MgBr) to yield tertiary alcohols. The first equivalent adds to form a ketone, and the second equivalent adds to the ketone to form the alcohol. This matches with option (T).
Reaction:
Final Answer:
The correct matches are:
First Reaction → (P)
Second Reaction → (Q)
Third Reaction → (R)
Fourth Reaction → (T)
Option (S) is not used.
1. Reduction Reactions:
- Nitro to Amine: Sn/HCl, Fe/HCl, Catalytic hydrogenation.
- Carboxylic Acid to Alcohol: LiAlH₄ is a standard reagent.
2. Nucleophilic Acyl Substitution:
This is the key reaction mechanism for acid chlorides and esters.
General Reaction:
Where L is a leaving group (Cl, OR').
3. Reactivity of Carbonyl Compounds:
The reactivity order is: Acid Chloride > Aldehyde > Ketone > Ester > Carboxylic Acid > Amide.
This explains why (CH₃)₂Cd (less reactive) stops at the ketone, while CH₃MgBr (more reactive) proceeds to the alcohol.