The major product of the following reaction is:
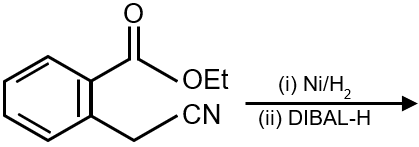
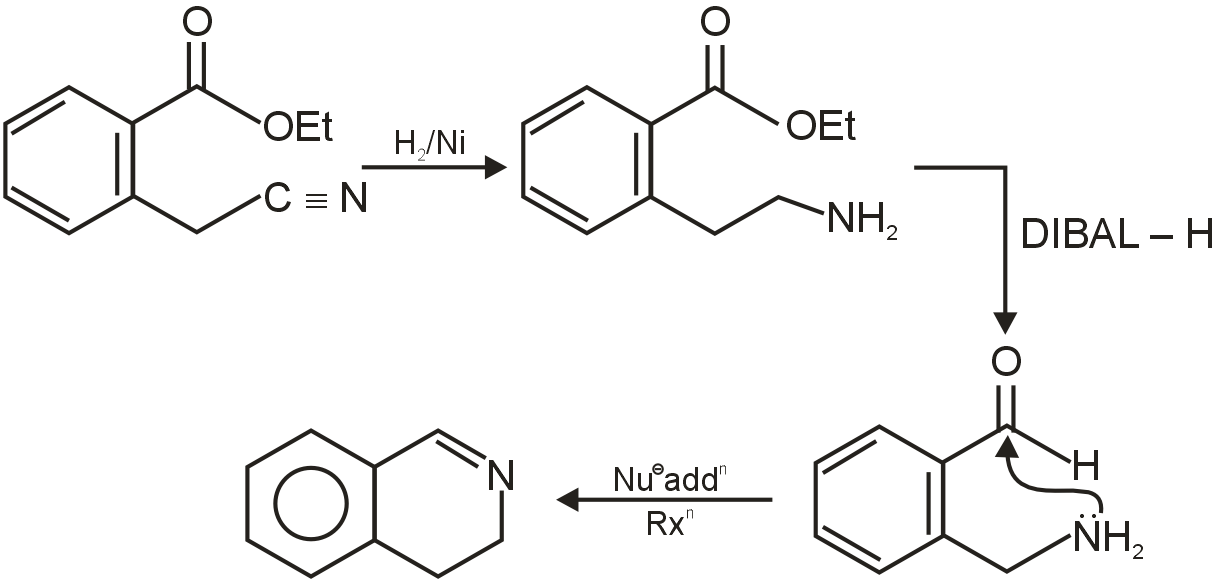
The given reaction involves an alkene (cyclopentene) reacting with diborane (B2H6) followed by oxidation with hydrogen peroxide (H2O2) in a basic medium (NaOH). This is a classic two-step process known as Hydroboration-Oxidation.
Step 1: Hydroboration
Diborane (B2H6) dissociates to form borane (BH3), which acts as the electrophile. The alkene undergoes syn addition across the double bond. The key feature of hydroboration is that it occurs with anti-Markovnikov regiochemistry and involves a syn addition (both new bonds form on the same face of the double bond).
For cyclopentene, the addition is not regioselective because the molecule is symmetric; both carbons of the double bond are equivalent. The product of this step is an alkylborane, trialkylborane.
Reaction:
Step 2: Oxidation
The alkylborane is oxidized by hydrogen peroxide in a basic solution (NaOH). The base deprotonates H2O2 to form the hydroperoxide ion (HOO-), which attacks the boron atom. This step replaces the boron group with a hydroxyl group (), with retention of configuration.
The overall result is the addition of water (H-OH) across the double bond, but in an anti-Markovnikov fashion and with syn stereochemistry.
Reaction:
OH
Final Answer:
For a symmetric alkene like cyclopentene, the hydroboration-oxidation reaction yields the corresponding alcohol where the OH group is attached to one carbon and the H is attached to the other. Since the alkene is symmetric, the product is cyclopentanol. The correct structure is a cyclopentane ring with an -OH group attached.
This corresponds to the first option: 
Hydroboration-Oxidation Overall Reaction:
B2H6 OH
Key Theory: The reaction proceeds via a four-membered transition state, which dictates the anti-Markovnikov addition (the boron atom bonds to the less substituted carbon) and syn stereochemistry. The oxidation step occurs with retention of configuration at the carbon.