The major product of the following reaction is
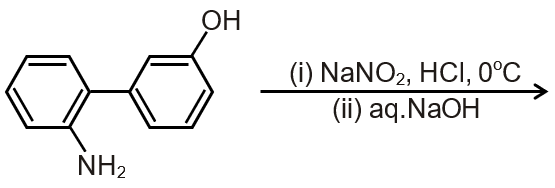
This reaction involves an aromatic compound (benzene ring) reacting with an alkyl halide in the presence of a Lewis acid catalyst (AlCl₃). This is a classic example of Friedel-Crafts alkylation, a type of electrophilic aromatic substitution.
Step 1: Generation of the Electrophile
The Lewis acid AlCl₃ coordinates with the chlorine atom of the alkyl chloride (CH₃-CH₂-Cl), polarizing the C-Cl bond. This leads to the formation of a carbocation electrophile.
The electrophile generated is an ethyl carbocation (CH₃CH₂⁺).
Step 2: Electrophilic Attack
The strong electrophile (CH₃CH₂⁺) attacks the electron-rich benzene ring. This forms a resonance-stabilized carbocation intermediate known as a sigma complex.
Step 3: Loss of a Proton
A base (often the [AlCl₄]⁻ ion generated in step 1) deprotonates the sigma complex. This restores the aromaticity of the benzene ring and yields the final substituted product: ethylbenzene.
Final Answer: The major product is Ethylbenzene. Among the given options, it is the structure with a benzene ring bonded to a -CH₂CH₃ group (the first option).
1. Electrophilic Aromatic Substitution (EAS): A reaction where an atom (usually H) attached to an aromatic ring is replaced by an electrophile. The general mechanism involves two main steps: (a) electrophilic attack to form a sigma complex and (b) loss of a proton.
2. Friedel-Crafts Reactions: A subset of EAS reactions that use Lewis acid catalysts to create powerful electrophiles.
3. Limitations of Friedel-Crafts Alkylation: