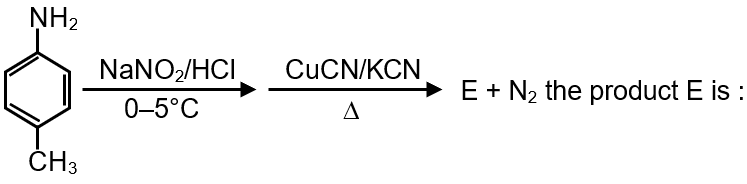
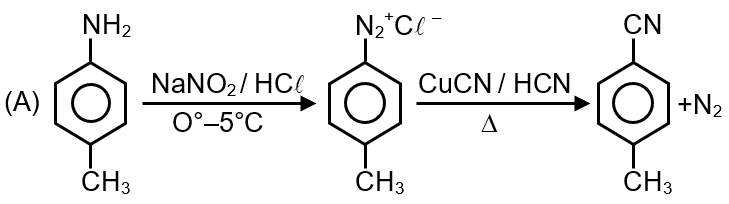
In the reaction :
This question involves an organic reaction where a compound undergoes a transformation, and we need to identify the correct major product from the given options. The reaction likely involves concepts like carbocation stability, rearrangement, or stereochemistry.
Step 1: Analyze the Starting Material
The starting material is a bicyclic compound with a bromine atom attached to a tertiary carbon. This suggests it is a tertiary alkyl halide.
Step 2: Understand the Reaction Conditions
The reaction is treated with a base (e.g., NaOH) or under conditions that promote elimination (E2) or substitution (SN1/SN2). Given it's a tertiary halide, it is more likely to undergo E2 elimination (with strong base) or SN1 substitution (with weak base/nucleophile). However, the presence of a base like NaOH typically favors elimination for tertiary halides.
Step 3: Predict the Mechanism
For a tertiary alkyl halide with a strong base (e.g., NaOH), E2 elimination is favored. The base abstracts a β-hydrogen, leading to the formation of an alkene. The major product is the most stable alkene following Zaitsev's rule (more substituted alkene is favored).
Step 4: Consider Stereochemistry and Rearrangement
In bicyclic systems, stereoelectronic factors and ring strain might influence the product. The elimination might give an exocyclic double bond or endocyclic double bond depending on stability. No rearrangement is expected in E2 as it is concerted.
Step 5: Compare with Options
Look for the option that represents the most stable alkene. Typically, it is the alkene with more substitution (tetrasubstituted > trisubstituted > disubstituted etc.). Also, in bridged systems, the double bond might be formed at the less strained position.
Final Answer: Based on stability, the major product is the trisubstituted alkene, which is option: (The image with the structure showing the double bond in the ring without bridgehead violation).
Zaitsev's Rule: In elimination reactions, the more substituted alkene is the major product.
E2 Mechanism: Bimolecular elimination, concerted process, rate = k[substrate][base].
Bredt's Rule: A double bond cannot be placed at the bridgehead of a bridged bicyclic system unless the ring is large enough.