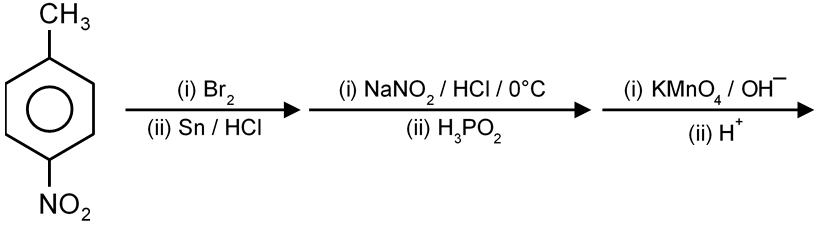
Final major product is
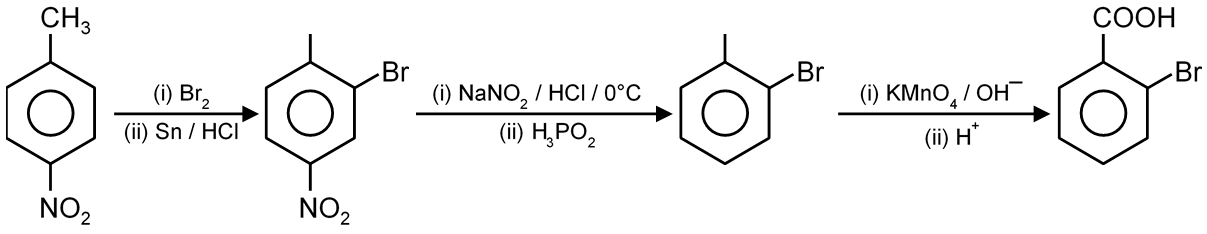
The given reaction involves the ozonolysis of an alkene. Ozonolysis is a reaction where an alkene is cleaved by ozone (O3) to form carbonyl compounds (aldehydes or ketones). The type of carbonyl compound produced depends on the substituents attached to the carbon atoms of the double bond.
Step 1: Analyze the Starting Alkene
The starting compound is a cyclic alkene. Let's interpret its structure from the first image. The molecule is 1-methylcyclopentene.
Structure: A cyclopentene ring with a methyl group () attached to one of the carbon atoms of the double bond.
Step 2: Understand Ozonolysis Mechanism
In ozonolysis, the carbon-carbon double bond is broken. Each carbon that was part of the double bond becomes a carbon in a new carbonyl group ().
Step 3: Apply the Rule to 1-Methylcyclopentene
Let's label the carbon atoms of the double bond in 1-methylcyclopentene as Carbon A and Carbon B.
Therefore, cleaving the double bond will produce two fragments: one ketone and one aldehyde.
Step 4: Predict the Carbonyl Products
The cleavage yields a single molecule because the alkene is cyclic. The product will be a dicarbonyl compound.
Joining these fragments together gives the final product: 5-oxohexanal. This is a 6-carbon chain with an aldehyde at one end () and a ketone at the 5th carbon (). Its structure is O=CH-(CH2)3-C(O)CH3.
Step 5: Match the Product to the Options
Now, let's compare our predicted structure (5-oxohexanal) to the provided options. The correct structure is the one that shows a 6-carbon chain with an aldehyde group on the left and a ketone group (C=O) on the fifth carbon, connected to a methyl group.
This matches the third option.
Final Answer: The third option is the correct major product.
Key Theory: Ozonolysis is a pivotal reaction for determining the position of a double bond in an alkene. The products are always carbonyl compounds, and their identities reveal the structure of the original alkene.
General Reaction:
Alkene + O3 → Carbonyl compounds (Aldehydes/Ketones)
Predicting Products:
Mechanism: The reaction proceeds via the formation of an unstable intermediate called an ozonide, which is then reduced (typically with Zn/AcOH or dimethyl sulfide) to yield the stable carbonyl compounds. The reductive workup prevents further oxidation of aldehydes to carboxylic acids.