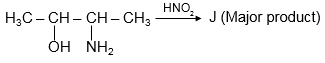
Which statement is incorrect about above major product ?
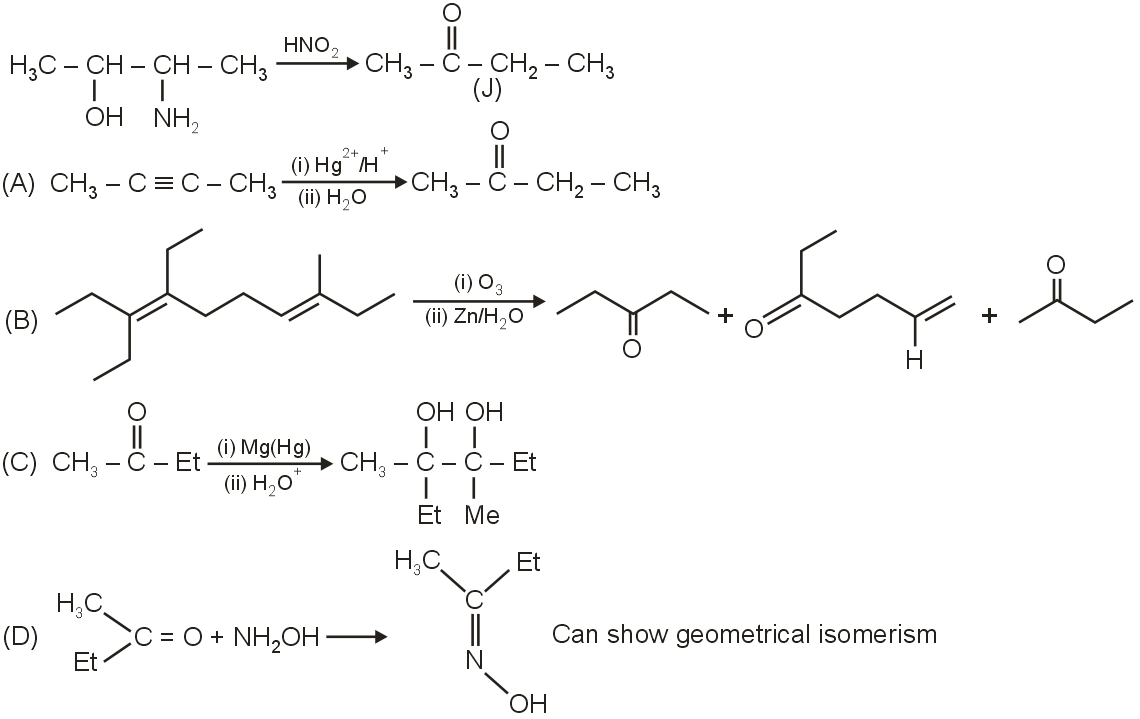
The given image shows a chemical structure, which is identified as Butanone (CH3COCH2CH3). We need to determine which statement about its major product or reactions is incorrect.
Butanone is a ketone. Reaction with hydroxylamine (NH2OH) forms an oxime. Butanone is unsymmetrical but the carbonyl carbon is not part of a carbon-carbon double bond; it is part of a ketone group. The oxime formed, CH3C(NOH)CH2CH3, does not have a C=N bond with two different substituents on the carbon atom that would allow for geometric (E/Z) isomerism. Therefore, this statement is incorrect.
The given alkene is 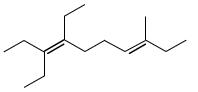 , which is 3,4-Dimethylhex-3-ene. Ozonolysis of this alkene would cleave the double bond. The products would be CH3COCH3 (acetone) and CH3CH2COCH3 (butanone). So, butanone is indeed a product. This statement is correct.
, which is 3,4-Dimethylhex-3-ene. Ozonolysis of this alkene would cleave the double bond. The products would be CH3COCH3 (acetone) and CH3CH2COCH3 (butanone). So, butanone is indeed a product. This statement is correct.
Assuming "J" refers to Butanone, the reaction described is Clemmensen reduction (Zn(Hg)/HCl) or similar, which reduces ketones to alkanes. For Butanone (CH3COCH2CH3), reduction would give Butane (CH3CH2CH2CH3). The given product is  , which is Cyclopropane. This is not a product of Butanone reduction. This statement is likely incorrect, but since Option 1 is already incorrect, we need to check all.
, which is Cyclopropane. This is not a product of Butanone reduction. This statement is likely incorrect, but since Option 1 is already incorrect, we need to check all.
But-2-yne (CH3C≡CCH3) is a symmetrical alkyne. Hydration with Hg2+ and acid gives ketones via keto-enol tautomerism. For symmetrical alkynes, it gives a single ketone: Butanone (CH3COCH2CH3). This statement is correct.
Both Option 1 and Option 3 seem incorrect. However, Option 1 is definitively incorrect because Butanone oxime does not show geometrical isomerism. Option 3 might be a trick, but typically "J" is Butanone, and reduction does not give cyclopropane. But since the question asks for the incorrect statement, and Option 1 is clearly wrong, it is the primary incorrect statement.
The incorrect statement is: "It can form geometrically isomers oxime on reaction with NH2–OH".
Oxime Formation: (Geometrical isomerism occurs if R and R' are different and the oxime has C=N bond with different groups)
Ozonolysis: Cleaves alkene double bonds to carbonyl compounds.
Clemmensen Reduction:
Alkyne Hydration: