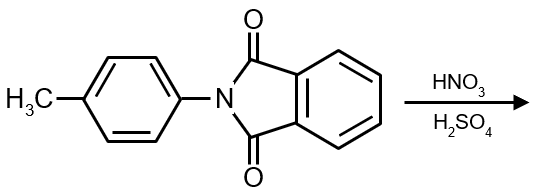
Product :
No solution
This question involves a nucleophilic substitution reaction where a nucleophile (CN⁻) attacks an alkyl halide substrate. The reaction follows SN2 mechanism due to the primary alkyl halide structure.
Step 1: Identify the substrate and nucleophile
Substrate: (1-bromopropane)
Nucleophile: (cyanide ion)
Step 2: Determine the reaction mechanism
Since the substrate is a primary alkyl halide, it undergoes SN2 reaction (bimolecular nucleophilic substitution).
Step 3: Understand stereochemistry
SN2 reactions proceed with inversion of configuration. The nucleophile attacks from the backside, causing the stereochemistry to flip.
Step 4: Predict the product
The cyanide ion replaces the bromine atom, forming: (butanenitrile)
Final Answer: The correct product is the second option showing
SN2 Rate Equation:
Key Characteristics of SN2: