This transformation involves the reduction of a nitrile group (-C≡N) to a primary amine (-CH2-NH2). Let's analyze the reagents:
Step 1: Understanding the Functional Group Change
The reactant is a nitrile (R-CN), and the product is a primary amine (R-CH2NH2). This requires reduction of the triple bond and addition of hydrogen.
Step 2: Evaluating Each Reagent
- Raney Ni/H2 in THF: This is a catalytic hydrogenation method. It reduces nitriles to primary amines: . It works.
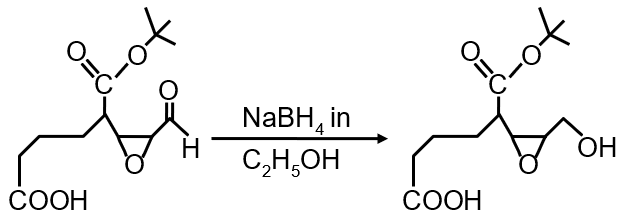
- NaBH4 in C2H5OH: Sodium borohydride is a mild reducing agent. It does not reduce nitriles to amines. It is ineffective for this transformation.
- LiAlH4 in (C2H5)2O: Lithium aluminium hydride is a strong reducing agent. It reduces nitriles to primary amines: . It works.
- BH3 in THF: Borane (often used as BH3·THF complex) is also a reducing agent. However, it is not commonly used for the reduction of nitriles. It is more selective for reducing carboxylic acids and is ineffective for nitriles.
Final Answer: The reagents which can be used are Raney Ni/H2 in THF and LiAlH4 in (C2H5)2O.
Related Topics & Formulae
Reduction of Nitriles: A key reaction in organic synthesis to prepare primary amines. The general reaction is:
Strength of Reducing Agents:
- LiAlH4 (Strong): Reduces carbonyls, carboxylic acids, esters, nitriles, etc.
- NaBH4 (Mild): Reduces aldehydes and ketones, but not acids, esters, or nitriles.
- Catalytic Hydrogenation (e.g., H2/Ni): Reduces alkenes, alkynes, nitro groups, and nitriles.
- BH3 (Selective): Preferentially reduces carboxylic acids over other carbonyls; does not reduce nitriles.