In the given transformation, which of the following is the most appropriate reagent ?
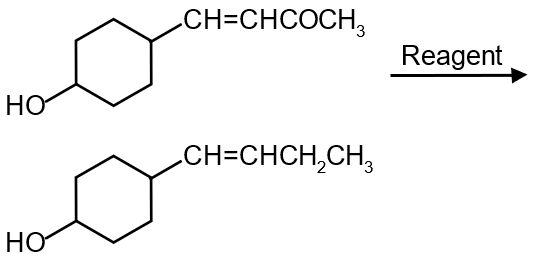
Reagent should not effect – OH and C=C
Alkaline medium is best suited
Wolf - Khischner is most appropriate.
This question involves identifying the correct reagent for reducing an alkyne to a trans-alkene. The transformation shown is from an alkyne (R-C≡C-R') to an alkene with the hydrogens on opposite sides (trans configuration).
The key concept here is stereoselective reduction. Different reducing agents give different stereochemical outcomes:
The dissolving metal reduction uses alkali metals (Na or Li) in liquid ammonia (NH3). This reaction proceeds via a radical anion mechanism that favors the formation of the more stable trans-alkene.
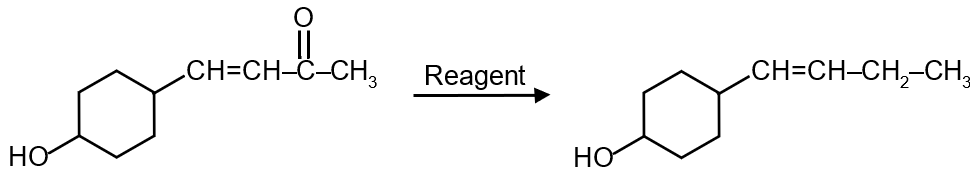
The complete reaction is:
Now, let's evaluate the options:
Therefore, the correct reagent is Na, Liq. NH3.
Reduction Reactions in Organic Chemistry: Different functional groups require specific reducing agents. Understanding the selectivity of reagents (which groups they reduce and the stereochemistry) is crucial in organic synthesis.
Alkyne Chemistry: Alkynes can undergo addition reactions (hydrogenation, halogenation, hydrohalogenation) and can be reduced to alkenes or alkanes with control over stereochemistry.
Dissolving Metal Reduction:
Catalytic Hydrogenation to cis-Alkene: