Considering the basic strength of amines in aqueous solution, which one has the smallest pkb value?
Basic strength of amine is sol.
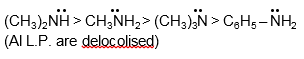
The basic strength of amines in aqueous solution depends on two main factors: the electron-donating inductive effect of alkyl groups and the stability of the conjugate acid formed after protonation. A smaller pKb value indicates a stronger base.
Step 1: Understand pKb and basic strength
pKb is the negative logarithm of the base dissociation constant (Kb). A smaller pKb means a larger Kb, which means the base is stronger.
Step 2: Compare alkyl amines
Alkyl groups are electron-donating, which increases the electron density on the nitrogen atom, making it a stronger base. Among simple alkyl amines:
The order of basic strength in water is: secondary > primary > tertiary.
Step 3: Consider aromatic amine
Aniline (C6H5NH2) has a phenyl group attached to the nitrogen. The phenyl group is electron-withdrawing due to resonance, which decreases the electron density on nitrogen, making it a very weak base. It has a much larger pKb compared to alkyl amines.
Step 4: Final comparison
Among the given options:
Basic Strength and pKb:
The base dissociation constant Kb for an amine B is given by: pKb = -log10(Kb). A smaller pKb means a stronger base.
Factors Affecting Basicity:
- Inductive Effect: Electron-donating groups (like alkyl groups) increase basicity; electron-withdrawing groups (like phenyl) decrease it.
- Solvation Effect: In water, the conjugate acid of primary amines is more solvated than tertiary amines due to more hydrogen bonding.
- Steric Hindrance: Bulky groups around nitrogen make it harder to protonate, reducing basicity.
Order in Aqueous Solution:
For alkyl amines: (CH3)2NH > CH3NH2 > (CH3)3N > NH3
Aromatic amines are much weaker: C6H5NH2 has pKb ~ 9.38, while (CH3)2NH has pKb ~ 3.27.