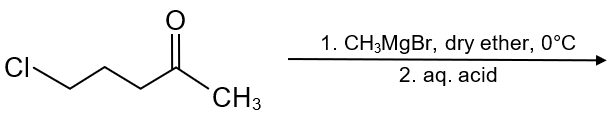
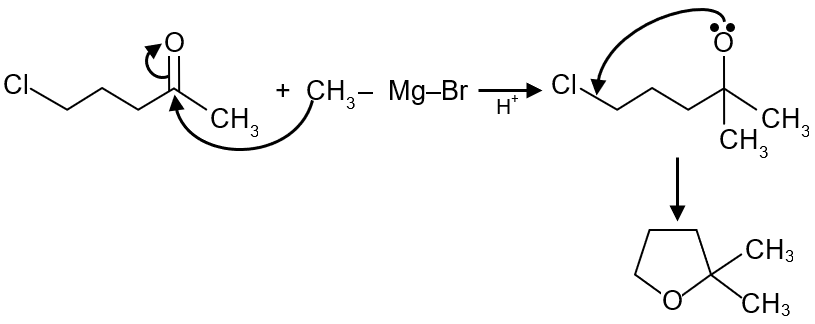
The major product in the following reaction is
The given reaction involves a conjugated diene (1,3-butadiene derivative) undergoing electrophilic addition with HBr. The key concept is 1,2-addition vs 1,4-addition under kinetic vs thermodynamic control.
The reactant is a substituted 1,3-butadiene: 2-methylpenta-1,3-diene. Its structure is:
CH2=C(CH3)–CH=CH–CH3
The electrophile is HBr (hydrogen bromide).
HBr adds to the conjugated diene via electrophilic addition. The first step is the attack of the electrophile (H+) on the diene system.
Formation of Allylic Carbocation Intermediate: The proton (H+) can add to either terminal carbon (C1 or C4) of the diene. Adding to C1 (the less substituted end) forms a more stable, tertiary allylic carbocation.
Protonation at C1:
This generates a resonance-stabilized tertiary allylic carbocation.
The positive charge is delocalized over two carbons (C2 and C4).
Structure I (Positive charge on C2):
Structure II (Positive charge on C4):
Structure I is tertiary, making it the major contributor to the hybrid.
The bromide ion (Br-) can attack either of the two carbocations (C2 or C4) from the resonance hybrid.
At low temperatures, the reaction is under kinetic control and the 1,2-adduct is the major product. However, this reaction is typically conducted at room temperature or above, favoring the thermodynamic product (1,4-adduct).
The major product is the more stable 1,4-addition product: 4-bromo-2-methylpent-2-ene.
Its structure is: BrCH2–C(CH3)=CH–CH2CH3
This matches the structure in the second option.
The major product is the 1,4-addition product: 4-bromo-2-methylpent-2-ene.
General Reaction: Conjugated diene + HX → 1,2-adduct + 1,4-adduct
Stability of Carbocations: Tertiary > Secondary > Primary > Methyl
Resonance Energy: Allylic systems are stabilized by resonance (10-15 kcal/mol).
Kinetic Control: The product that forms fastest (lower activation energy) is favored at low temperatures. This is usually the 1,2-adduct.
Thermodynamic Control: The most stable product is favored at higher temperatures and longer reaction times. This is usually the 1,4-adduct with a more substituted alkene.