Match the reactions in column I with appropriate types of steps / reactive intermediate involved in these reactions as given in column II
| Column-I | Column-II |
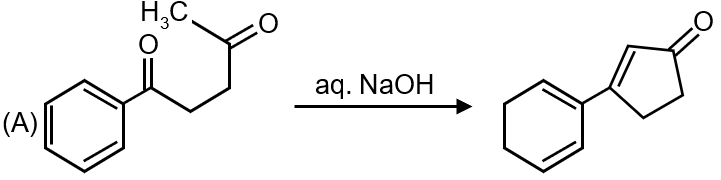 |
(p) Nucleophilic substitution |
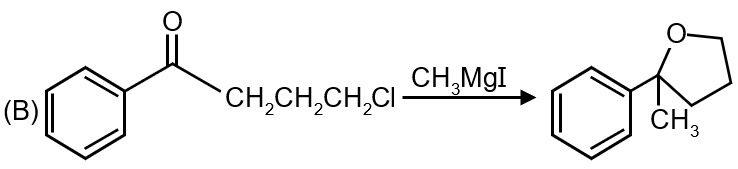 |
(q) Electrophilic substitution |
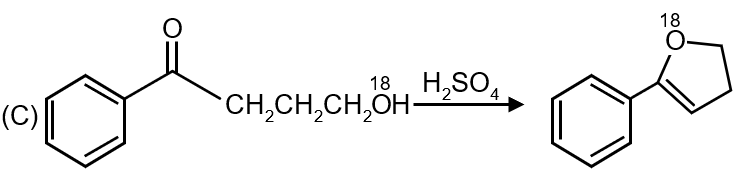 |
(r) Dehydration |
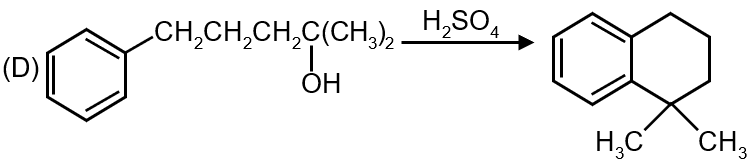 |
(s) Nucleophilic addition |
| (t) Carbanion |
Factual
* The most appropriate answer to this question is
A r, s, t; B p, s, t; C r, s; D q, r
But because of ambiguity in language, IIT has declared
A r, s, t; B p, s; C r, s; D q & r as correct answer
This question involves matching organic reactions with their mechanism types and reactive intermediates. Let's analyze each reaction step by step:
The first reaction shows a carbonyl compound undergoing addition of HCN to form a cyanohydrin. This is a classic example of nucleophilic addition where CN⁻ acts as a nucleophile attacking the electrophilic carbon of the carbonyl group. The intermediate formed is a carbanion-like species during the reaction.
Matches: (s) Nucleophilic addition, (t) Carbanion
The second reaction shows an alkyl halide converting to an alcohol using aqueous KOH. This is a classic nucleophilic substitution reaction (SN2 or SN1) where OH⁻ replaces the halogen atom.
Matches: (p) Nucleophilic substitution
The third reaction shows an alcohol converting to an alkene using concentrated H2SO4. This is dehydration (elimination reaction) where water is removed from the alcohol molecule.
Matches: (r) Dehydration
The fourth reaction shows nitration of benzene using HNO3/H2SO4. This is a classic electrophilic substitution reaction where NO2+ (nitronium ion) acts as the electrophile.
Matches: (q) Electrophilic substitution
Nucleophilic Addition: Characteristic of carbonyl compounds (aldehydes/ketones) where nucleophiles add to the electrophilic carbon.
Nucleophilic Substitution: Halogen atoms in alkyl halides are replaced by nucleophiles like OH⁻, CN⁻, etc.
Dehydration: Removal of water molecule from alcohols to form alkenes, typically acid-catalyzed.
Electrophilic Substitution: Aromatic compounds undergo substitution where electrophiles replace hydrogen atoms.
Carbanion: A reactive intermediate with a negatively charged carbon atom, often formed in nucleophilic addition reactions.
Reaction 1 → (s) and (t)
Reaction 2 → (p)
Reaction 3 → (r)
Reaction 4 → (q)