2–Hexyne gives trans-2-Hexene on treatment with :
This question involves understanding the selective reduction of alkynes to alkenes using specific reagents. The key concept is that different catalysts/reagents give different stereochemical outcomes (cis or trans alkenes).
An alkyne (like 2-hexyne) can be reduced to an alkene (2-hexene) via catalytic hydrogenation. However, the stereochemistry of the product depends on the reagent used:
Step 1: Catalytic Hydrogenation with Pt or Pd
Common catalysts like Pt or Pd (e.g., Pt/H2 or Pd/C) give complete reduction to the alkane. They are too reactive and do not stop at the alkene stage. Even Pd/BaSO4 (Lindlar's catalyst) is used for reduction, but it gives the cis-alkene, not the trans isomer.
Step 2: Reduction with LiAlH4
LiAlH4 is a strong reducing agent used primarily for reducing carbonyl groups, carboxylic acids, etc. It is not generally used for reducing carbon-carbon triple bonds. It would not give the desired alkene product.
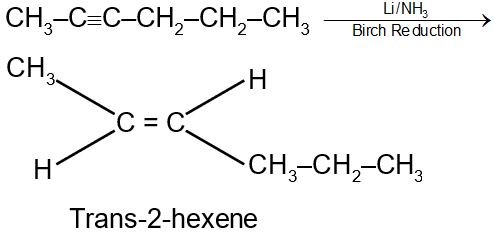
Step 3: Reduction with Li/NH3 (lithium in liquid ammonia)
This is a dissolving metal reduction. The mechanism involves single electron transfers, forming a radical anion intermediate. The trans geometry is favored due to steric reasons in the intermediate. The reaction is:
Thus, Li/NH3 is the reagent that selectively reduces an internal alkyne to the trans-alkene.
Final Answer: The correct reagent is Li/NH3 (lithium in liquid ammonia).
Alkyne Reduction Reactions:
Mechanism Insight: The dissolving metal reduction (Li/NH3) proceeds via a radical anion mechanism. An electron is transferred from Li to the alkyne, forming a radical anion. This anion is protonated by NH3, and another electron transfer followed by protonation gives the trans-alkene due to the anti-addition.