Match the reactions in Column I with appropriate options in Column II.
| Column-I | Column-II |
| (p) Racemic mixture | |
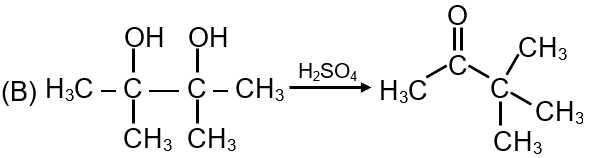 |
(q) Addition reaction |
 |
(r) Substitution reaction |
| (s) Coupling reaction | |
|
(t) Carbocation intermediate |
No solution
This question involves matching organic reactions from Column I with their correct descriptions in Column II. Let's analyze each reaction step by step:
The first reaction shows a carbonyl compound (likely an aldehyde or ketone) reacting with HCN. This is a nucleophilic addition reaction where CN⁻ attacks the electrophilic carbon of the carbonyl group. The product is a cyanohydrin. Since the carbonyl carbon is planar (sp² hybridized), the nucleophile can attack from either face, leading to a racemic mixture if the carbonyl compound is unsymmetrical. This reaction proceeds via a carbocation intermediate is not typical; instead, it goes through a tetrahedral intermediate.
Matches: (p) Racemic mixture and (q) Addition reaction
The second reaction involves benzene with Cl₂ in the presence of anhydrous AlCl₃. This is a classic electrophilic aromatic substitution reaction, specifically chlorination. The AlCl₃ acts as a Lewis acid to generate the electrophile Cl⁺, which attacks the aromatic ring. Substitution occurs, replacing a hydrogen with chlorine. No carbocation intermediate is involved in the typical sense; instead, a sigma complex (arenium ion) is formed.
Matches: (r) Substitution reaction
The third reaction shows a diazonium salt (Ar-N₂⁺) reacting with phenol in basic medium. This is a diazonium coupling reaction, which is an electrophilic substitution where the diazonium cation acts as the electrophile, attacking the electron-rich aromatic ring of phenol. The product is an azo compound. This reaction does not involve a racemic mixture, addition, or carbocation intermediate.
Matches: (s) Coupling reaction
The fourth reaction is an alkene reacting with HBr. This is an electrophilic addition reaction following Markovnikov's rule. The mechanism involves the formation of a carbocation intermediate: the π electrons of the alkene attack H⁺ from HBr, forming a carbocation, which is then attacked by Br⁻. If the alkene is unsymmetrical, the carbocation stability dictates the major product. This reaction does not produce a racemic mixture unless the carbocation is planar and attacked from both sides, but typically for unsymmetrical alkenes, it gives a racemic mixture if the carbocation is chiral.
Matches: (q) Addition reaction and (t) Carbocation intermediate
Nucleophilic Addition: Reactions where a nucleophile adds to a carbonyl group, common in aldehydes and ketones. Example: addition of HCN to form cyanohydrins.
Electrophilic Aromatic Substitution: Reactions where an electrophile substitutes a hydrogen on an aromatic ring, e.g., halogenation, nitration.
Diazonium Coupling: A reaction where diazonium salts react with electron-rich aromatics to form azo compounds, important in dye chemistry.
Electrophilic Addition: Addition reactions to alkenes where an electrophile adds first, forming a carbocation intermediate, followed by nucleophile addition.
General Reaction for Nucleophilic Addition to Carbonyl:
Electrophilic Aromatic Substitution Mechanism: Formation of electrophile followed by attack and deprotonation.
Markovnikov's Rule: In addition of HX to unsymmetrical alkenes, H adds to the carbon with more hydrogens.