Amongst the compounds given, the one that would form a brilliant colored dye on treatment with NaNO2 in dil. HCl followed by addition to an alkaline solution of β‑naphthol is :
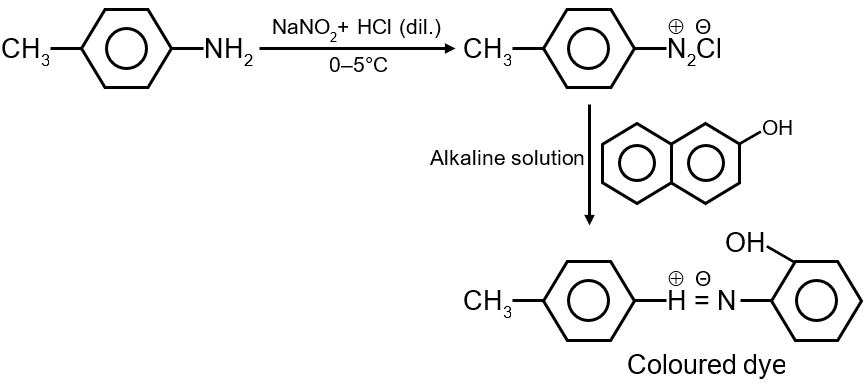
This question involves identifying which compound forms a colored azo dye when treated with NaNO2 and dilute HCl (diazotization) followed by coupling with β-naphthol in alkaline solution. This is a classic test for aromatic primary amines to form azo dyes.
Key Concept: Only aromatic primary amines (Ar-NH2) undergo diazotization to form diazonium salts (Ar-N2+Cl-). These diazonium salts then couple with electron-rich aromatic compounds like β-naphthol (in alkaline medium) to form brightly colored azo dyes.
Let's analyze the given options (structures not fully visible, but based on typical patterns):
The correct compound must contain an aromatic ring with a primary amino group (-NH2) directly attached. Only such compounds undergo this reaction sequence.
Reaction Steps:
Step 1: Diazotization
Step 2: Coupling with β-naphthol (in NaOH)
Final Answer: The compound with the aromatic primary amine group (option 1, based on typical structures) is correct.
Diazotization: Conversion of primary aromatic amines to diazonium salts using NaNO2 and acid.
Azo Coupling: Electrophilic substitution where diazonium salt attacks electron-rich aromatics like phenols/anilines.
β-naphthol: Commonly used coupling agent due to high electron density.
Important Note: Aliphatic amines do not form stable diazonium salts. Only aromatic primary amines give this test.