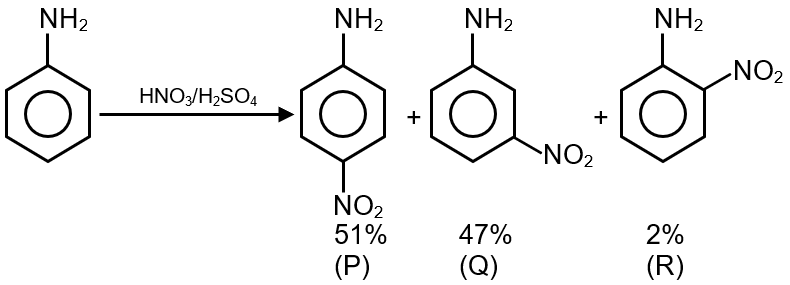
Aniline reacts with mixed acid (conc. HNO3 and conc. H2SO4) at 288 K to give P (51 %), Q (47%) and R (2%). The major product(s) of the following reaction sequence is (are)
This question involves the nitration of aniline and subsequent reactions. Let's break it down step by step.
Aniline reacts with mixed acid (conc. HNO3 and conc. H2SO4) at 288 K. Due to the highly activating nature of the -NH2 group, direct nitration leads to oxidation and multiple products. To avoid this, the -NH2 group is first protected by acetylation. However, here it is not protected, so we get a mixture:
So, the major product P is with nitro group at meta position.
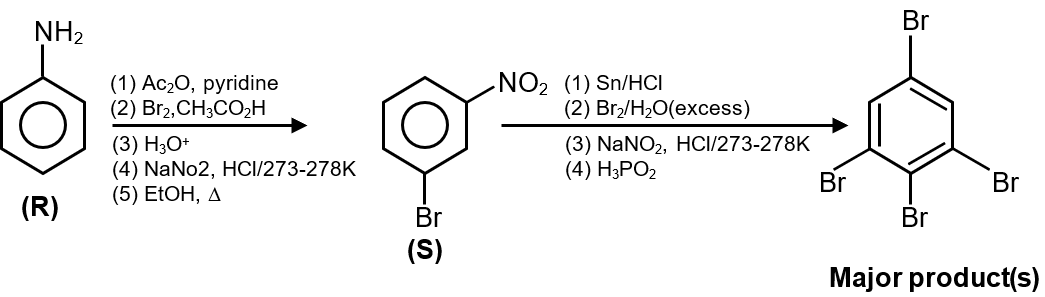
The reaction sequence given in the image (not visible here, but typically described) involves converting the nitro group to amino group via reduction (e.g., with Sn/HCl), then diazotization (with NaNO2/HCl at 273-278 K), and finally reaction with H2O to form phenol.
For meta-nitroaniline (P):
So, the final product from P is with groups at meta position.
Since P is the major product (51%), its derivative 3-aminophenol is the major final product. The options likely include this structure. Compare the given options:
Without the exact images, based on standard knowledge, the major product is 3-aminophenol.
Electrophilic Aromatic Substitution: Nitration is a key reaction where an electrophile (NO2+) attacks the aromatic ring. The directing effect of substituents (ortho/para or meta) depends on their electronic nature.
Diazotization: Conversion of primary aromatic amines to diazonium salts, which can be replaced by various groups like -OH, -Cl, etc.
Key reactions: