Ozonolysis of an organic compounds gives formaldehyde as one of the products. This confirms the presence of :
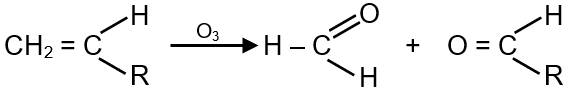
Presence of one vinyl group gives formaldehyde as one of the product in ozonolysis.
Ozonolysis is a reaction where ozone cleaves carbon-carbon double bonds (C=C) or triple bonds (C≡C) in organic compounds. When formaldehyde (HCHO) is produced as one of the products, it indicates the presence of a terminal alkene group, specifically a vinyl group.
Step 1: Understand Ozonolysis Reaction
Ozonolysis of alkenes involves cleavage of the double bond and formation of carbonyl compounds (aldehydes or ketones). The general reaction is:
Step 2: Analyze Formaldehyde Production
Formaldehyde (HCHO) is formed only when one end of the double bond has two hydrogen atoms, i.e., a terminal double bond (vinyl group: -CH=CH2). For example:
Step 3: Evaluate Other Options
- Acetylenic triple bond: Produces carboxylic acids, not formaldehyde.
- Two ethylenic double bonds: May not necessarily produce formaldehyde unless one is terminal.
- Isopropyl group: Not directly cleaved by ozonolysis; it is an alkyl group.
Final Answer: The presence of formaldehyde confirms a vinyl group (-CH=CH2).
General ozonolysis reaction for alkenes:
If R3 and R4 are H, then formaldehyde (HCHO) is produced.