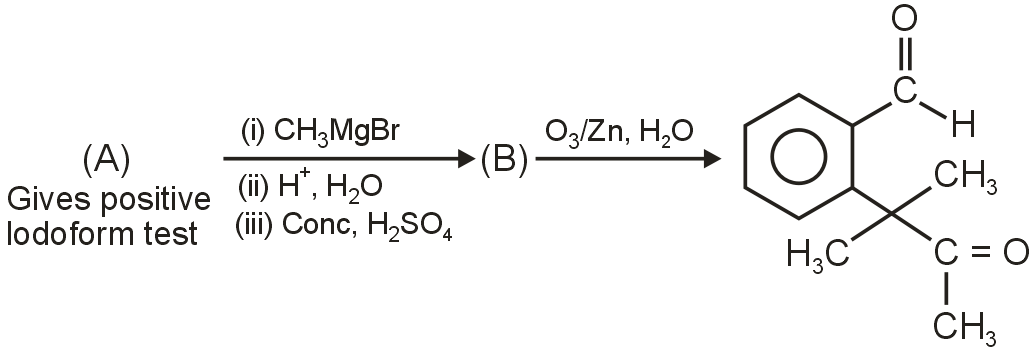
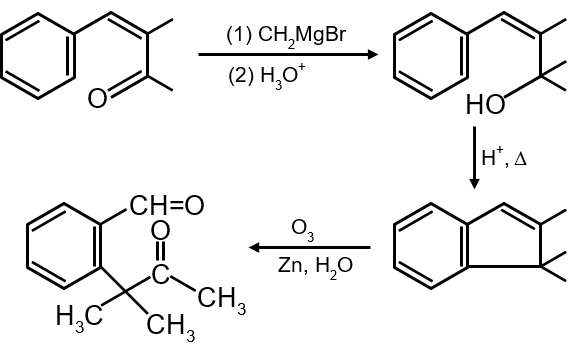
Identify (A) in the following reaction sequence :
The given reaction sequence involves a carboxylic acid derivative undergoing transformations. Let's analyze step by step:
The reaction begins with a carboxylic acid containing a β-carbonyl group (malonic acid derivative). The first reagent is PCl5, which converts -COOH to -COCl group.
The acid chloride then reacts with ammonia to form an amide through nucleophilic substitution:
The amide undergoes Hofmann degradation with Br2/NaOH to form an amine with one less carbon atom:
For malonic acid (HOOC-CH2-COOH), the sequence would produce glycine (H2N-CH2-COOH). Comparing with the options, the correct structure (A) is:
H2N-CH2-COOH (Glycine)
Acid Chloride Formation:
Amide Formation:
Hofmann Reaction: