Consider the given reactions :
Compound A undergoes polymerisation with hexamethylene diamine to form C.
Select the incorrect combination.
Let's analyze the given reactions step by step:
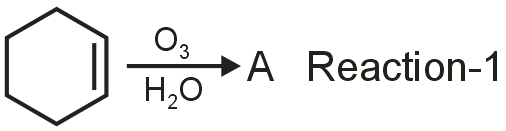
The first reaction shows ethylene undergoing ozonolysis. Ozonolysis of alkenes cleaves the double bond to form carbonyl compounds.
For ethylene (CH2=CH2):
This produces formaldehyde (HCHO). However, the problem mentions "oxidative ozonolysis" which typically uses H2O2 instead of Zn/H2O, converting aldehydes to carboxylic acids.
Ethylene reacts with Baeyer's reagent (cold alkaline KMnO4), which performs syn hydroxylation:
This forms ethylene glycol (a vicinal diol) through syn addition.
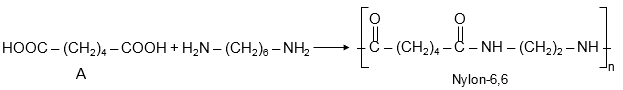
Compound A (from Reaction-1) polymerizes with hexamethylene diamine. Since hexamethylene diamine is H2N-(CH2)6-NH2, A must be a dicarboxylic acid to form a polyamide.
From oxidative ozonolysis of ethylene, we get formic acid (HCOOH), but this is monofunctional. However, if we consider a different alkene that gives a diacid upon oxidative ozonolysis, but here ethylene is given. There might be a misinterpretation.
Actually, for ethylene, oxidative ozonolysis would give formic acid (HCOOH), which is not suitable for polymerization with a diamine to form a polyamide. This suggests that A might not be from ethylene, or there's a different reactant.
But based on standard knowledge, when a diacid reacts with hexamethylene diamine, it forms Nylon-6,6, a step-growth polymer.
Now, evaluating each option:
The incorrect combination is: C–Dacron–Chain growth polymer
Ozonolysis cleaves alkenes at the double bond. General reaction:
With oxidative workup (H2O2), aldehydes are oxidized to carboxylic acids.
Syn addition of OH groups across the double bond:
Step-growth polymerization: Monomers react to form dimers, trimers, etc., gradually. Example: Nylon, polyester.
Chain-growth polymerization: Involves initiation, propagation, termination. Example: Polyethylene, PVC.