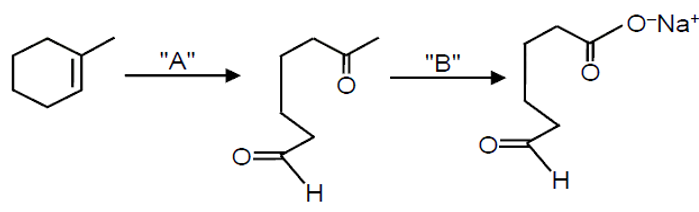
In the above chemical reaction sequence "A" and "B" respectively are
The given reaction involves a cyclic alkene undergoing ozonolysis (reaction with A) followed by a specific treatment (B) to form a dicarboxylic acid. Let's analyze step by step.
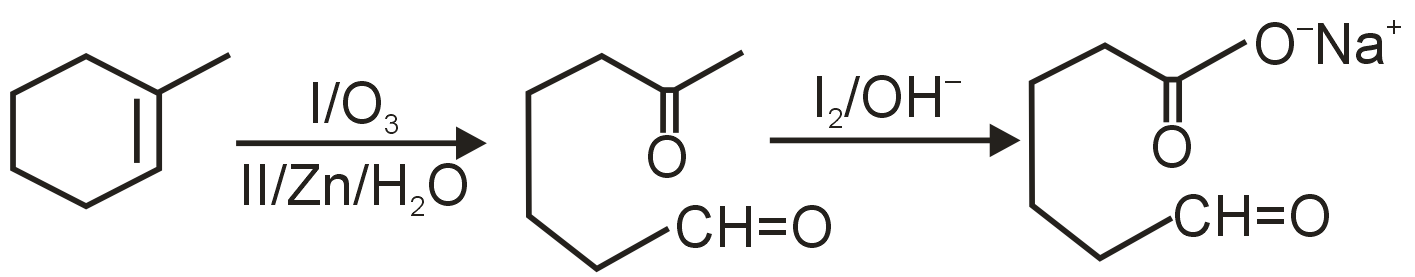
The first step converts the alkene into carbonyl compounds. Ozonolysis is used to cleave alkenes at the double bond. For an alkene, ozonolysis with O3 followed by Zn/H2O gives aldehydes or ketones. Here, the product after A is not shown, but the final product is a dicarboxylic acid, indicating that the intermediate carbonyls are aldehydes (which can be oxidized to acids). Thus, A is O3, Zn/H2O.
Reaction:
The intermediate from ozonolysis is likely an aldehyde (since it forms a carboxylic acid upon oxidation). To convert aldehyde to carboxylic acid, we need an oxidizing agent. KMnO4 (under acidic or basic conditions) is commonly used for this oxidation. NaOH(alc)/I2 is used for iodoform test or oxidation of methyl ketones, not for direct aldehyde oxidation to acid. Thus, B is KMnO4.
Reaction:
Therefore, A is O3, Zn/H2O and B is KMnO4. The correct option is: O3, Zn/H2O and KMnO4.
Key Equations:
Ozonolysis:
Oxidation: