Which of the following compounds will form significant amount of meta product during mono-nitration reaction?
This question tests your understanding of electrophilic aromatic substitution, specifically nitration, and how substituents on the benzene ring direct the incoming nitro group (NO2+) to the ortho, meta, or para positions.
The key concept is that substituents are classified as ortho-para directing activators, ortho-para directing deactivators, or meta directing deactivators. A "significant amount of meta product" will be formed primarily when the substituent on the ring is a meta-directing deactivator.
These groups are electron-withdrawing and destabilize the carbocation intermediate formed during the reaction. The meta-position is less electron-deficient compared to the ortho and para positions relative to the substituent, making it the favored site for attack. Common meta-directing groups include:
To solve this, you must identify which compound among the options has a substituent that falls into this meta-directing deactivator category.
Since the exact structures from the images are not provided in text, I will describe the common compounds these images typically represent and their directing effects.
Option 1: This image often represents Phenol (C6H5OH).
The -OH group is a strong ortho-para directing activator. It will produce primarily ortho and para nitration products. A significant meta product will NOT be formed.
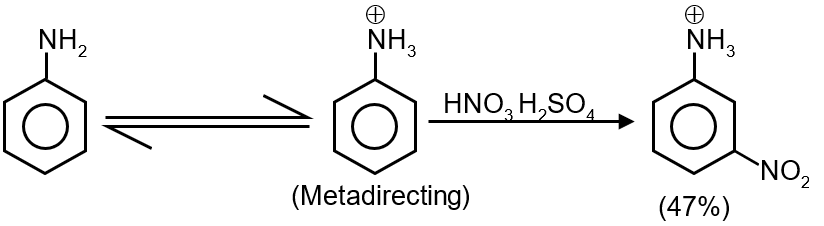
Option 2: This image often represents Aniline (C6H5NH2) or a similar compound.
The -NH2 group is a very strong ortho-para directing activator. It will also not produce a significant meta product.
Option 3: This image often represents Toluene (C6H5CH3).
The -CH3 group is an ortho-para directing activator (though weaker than -OH or -NH2). It directs substitution to the ortho and para positions.
Option 4: This image often represents Nitrobenzene (C6H5NO2) or Benzoic acid (C6H5COOH).
Both the -NO2 and -COOH groups are classic meta-directing deactivators. Nitration of these compounds occurs primarily at the meta position, yielding a significant amount of meta product.
Therefore, the compound in Option 4 (likely Nitrobenzene or Benzoic acid) is the one that will form a significant amount of meta product during mono-nitration.
Electrophilic Aromatic Substitution (EAS): The general mechanism for reactions like nitration, halogenation, sulfonation, and Friedel-Crafts alkylation/acylation. The rate and regioselectivity are governed by the nature of the substituent already present on the ring.
Directing Effects:
Resonance Theory: The directing effect is best explained by drawing the resonance structures of the carbocation (sigma complex) intermediate formed when the electrophile attacks the ortho, meta, and para positions. A meta-directing group ensures that no resonance structure places the positive charge on the carbon atom directly attached to it, which is highly destabilized.