The reactant that lead to product (X) and (Y) on ozonolysis are:
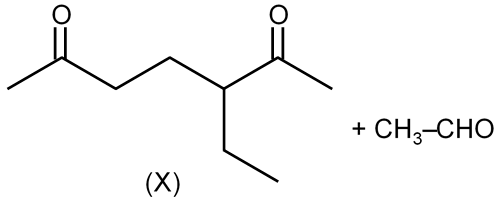
In ans, on ozonolysis give 'X'
Ozonolysis is a reaction where an alkene is cleaved by ozone (O3) to form carbonyl compounds (aldehydes or ketones). The process helps identify the structure of the original alkene by analyzing the products.
Given products (X) and (Y) from ozonolysis are:

This is OHC-CH2-CH2-CHO (a dialdehyde).
To find the reactant alkene, reverse the ozonolysis: combine the carbonyl carbons with a double bond. For a dialdehyde, the original alkene had a double bond between the two carbon atoms that became carbonyl groups.
Step 1: Identify the carbonyl carbons in (X). Both ends are aldehydes (CHO groups), so the double bond was between these two carbons. However, (X) has four carbons: the two terminal carbons are carbonyls, and the two middle are CH2 groups. This suggests the original alkene was symmetric.
Step 2: Reverse the reaction. The carbonyl carbons were part of the double bond. So, removing the oxygen atoms and connecting the carbons with a double bond gives the alkene.
For OHC-CH2-CH2-CHO, the carbonyl carbons are C1 and C4. Connecting C1 and C4 with a double bond would give CH2=CH-CH2-CH2? But that is not symmetric. Actually, since both ends are identical, the original alkene is cyclic or symmetric.
Actually, (X) is a straight chain dialdehyde. The reverse ozonolysis: each CHO group came from a =CH2 end. So, the original alkene should be H2C=CH-CH2-CH2-CH=CH2? But that would give two molecules of OHC-CH2-CH2-CHO? No, careful: ozonolysis of a diene might give multiple products.
Looking at the image, (X) is OHC-(CH2)2-CHO. This dialdehyde comes from an alkene that has a double bond between the two carbonyl carbons. So, the alkene is CH2=CH-CH2-CH2-CH=CH2? But that is not correct because it would cleave to two OHC-CH2-CH2-CHO molecules? Actually, it is a single molecule.
Actually, for a mono-alkene, ozonolysis gives two carbonyls. Here, (X) is one molecule with two aldehydes, meaning it came from a diene or a ring.
But the product is only one compound (X), so the reactant alkene must be cyclic. For a cyclic alkene, ozonolysis cleaves the ring and gives a dialdehyde.
So, for (X) OHC-(CH2)2-CHO, it comes from cyclopentene? Let's see: cyclopentene has 5 carbons. Ozonolysis would give OHC-(CH2)3-CHO, which is not matching.
OHC-(CH2)2-CHO has 4 carbons. So, it came from a cyclobutene derivative? Cyclobutene ozonolysis gives OHC-CH2-CH2-CHO, which is exactly (X).
Therefore, the reactant is cyclobutene.
Now, looking at the options:
Option 1:  This is cyclobutene.
This is cyclobutene.
Option 2:  This is cyclopentene.
This is cyclopentene.
Option 3:  This is 1,5-hexadiene.
This is 1,5-hexadiene.
Option 4: 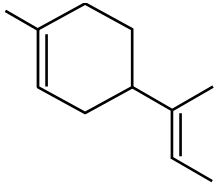 This is methylcyclobutene.
This is methylcyclobutene.
Only cyclobutene (Option 1) gives OHC-(CH2)2-CHO upon ozonolysis.
Ozonolysis of cyclobutene: the double bond is cleaved, and the ring opens to form OHC-CH2-CH2-CHO.
So, the correct reactant is Option 1.
Ozonolysis is part of alkene reactions. It involves the cleavage of double bonds to carbonyl compounds. Understanding the reverse process (working backwards from products to reactant) is key.
General ozonolysis reaction: (for terminal alkene)
For cyclic alkenes, it gives dialdehydes.