An organic compound neither reacts with neutral ferric chloride solution nor with Fehling solution. It however, reacts with Grignard reagent and gives positive iodoform test. The compound is :
Let's analyze the given clues step by step to identify the organic compound:
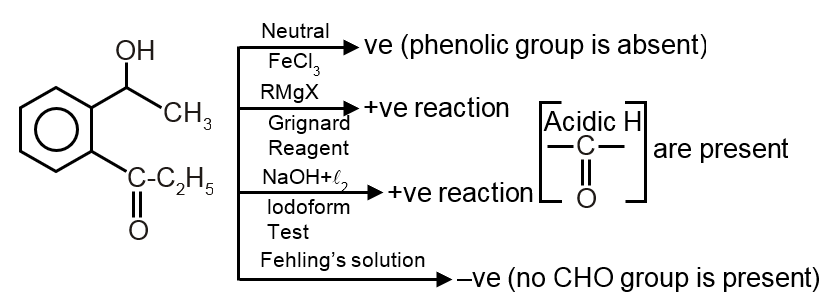
Step 1: Does not react with neutral ferric chloride solution
This test is positive for phenols (gives violet color) and enols. So, the compound is not a phenol.
Step 2: Does not react with Fehling solution
Fehling solution test is positive for aldehydes (except aromatic aldehydes) and reducing sugars. So, the compound is not an aliphatic aldehyde (aromatic aldehydes do not give this test).
Step 3: Reacts with Grignard reagent
Grignard reagent (RMgX) reacts with compounds having active hydrogen (like -OH, -COOH, -NH2) or carbonyl groups (aldehydes, ketones, esters, etc.). So, the compound likely contains a carbonyl group or active hydrogen.
Step 4: Gives positive iodoform test
Iodoform test is positive for:
- Methyl ketones (RCOCH3)
- Acetaldehyde (CH3CHO)
- Ethanol (CH3CH2OH)
- Secondary alcohols with methyl group adjacent to carbonyl (CH3CH(OH)R)
Since it does not react with Fehling (so not acetaldehyde) and not with neutral FeCl3 (so not enolizable phenols), it is likely a methyl ketone (RCOCH3).
Step 5: Combining all clues
The compound is a methyl ketone (positive iodoform) that is not an aliphatic aldehyde (negative Fehling) and not a phenol (negative FeCl3). It reacts with Grignard reagent (consistent with carbonyl group). So, it is a ketone with structure RCOCH3, where R is not a group that interferes with the tests.
Now, looking at the options (images not renderable here, but based on common structures):
- Option 1: Likely a phenol (would give positive FeCl3) – inconsistent.
- Option 2: Likely an aldehyde (may give Fehling test if aliphatic) – inconsistent.
- Option 3: Likely a methyl ketone (e.g., CH3COC6H5 or similar) – matches all criteria.
- Option 4: Likely an alcohol or non-methyl carbonyl – may not give iodoform test.
Thus, the compound is a methyl ketone, specifically one that is not an aldehyde or phenol. Common example is acetophenone (C6H5COCH3), which gives positive iodoform test, does not reduce Fehling (as it is ketone), does not react with neutral FeCl3 (not phenol), and reacts with Grignard reagent.
Final Answer: The compound is a methyl ketone, such as acetophenone.
Iodoform Test: Positive for compounds with CH3C=O group or CH3CH(OH)- group. Reaction:
Fehling Test: Positive for aliphatic aldehydes:
Neutral FeCl3 Test: Positive for phenols (violet color) and enols.
Grignard Reaction: (after hydrolysis).