The major product of the following reaction sequence is
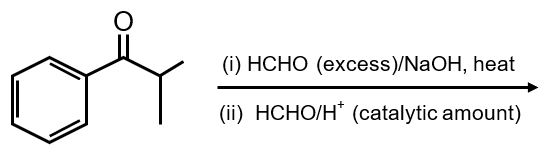
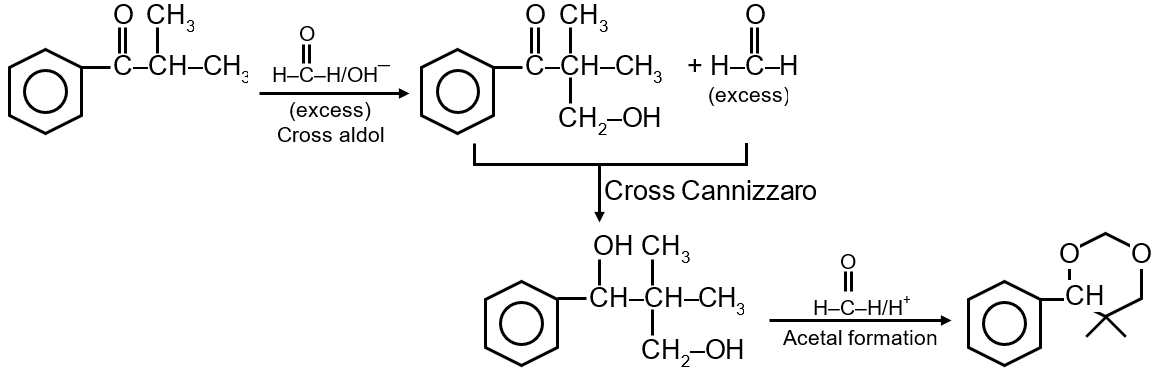
This reaction sequence involves a Grignard reagent addition followed by dehydration to form an alkene. Let's analyze it step by step:
Step 1: Grignard Reaction
The starting compound is a ketone (cyclopentanone). It reacts with ethyl magnesium bromide (CH3CH2MgBr), which is a Grignard reagent. Grignard reagents add to ketones to form tertiary alcohols. The reaction is:
Specifically, the product is 1-ethylcyclopentanol.
Step 2: Dehydration
The tertiary alcohol is treated with concentrated H2SO4 and heat. This causes dehydration, eliminating a water molecule (H2O) to form an alkene. Tertiary alcohols dehydrate easily via E1 mechanism, forming the most stable alkene according to Zaitsev's rule (more substituted alkene is favored).
In cyclopentanol derivatives, dehydration gives the alkene with the double bond outside the ring for stability, avoiding ring strain. Thus, the major product is ethylidenecyclopentane (1-ethylidenecyclopentane), which has the structure:
This is because the double bond is exocyclic to the ring, making it more stable than an endocyclic alkene.
Final Answer: The major product is the first option, ethylidenecyclopentane.
Grignard Reactions: Organometallic compounds like Grignard reagents (R-Mg-X) add to carbonyl groups (aldehydes, ketones) to form alcohols. With ketones, they yield tertiary alcohols.
Dehydration of Alcohols: Alcohols lose water under acidic conditions to form alkenes. The stability of the alkene depends on the degree of substitution (Zaitsev's rule). Tertiary alcohols dehydrate readily.
General Grignard addition to ketone:
Dehydration: