The hybridisation of orbitals of N atom in NO3–, NO2+ and NH4+ are respectively
Number of electron pairs = 2 Number of bond pairs = 2 Number of lone pair = 0 So, the species is linear with sp hybridisation
![]()
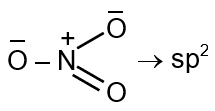
Number of electron pairs = 3 Number of bond pairs = 3 Number of lone pair = 0 So, the species is trigonal planar with sp2 hybridisation.
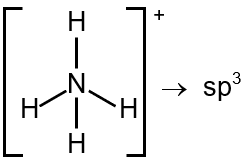
Number of electron pairs = 4 Number of bond pairs = 4 Number of lone pair = 0 So, the species is tetrahedral with sp3 hybridisation.
Hybridization is the concept of mixing atomic orbitals to form new hybrid orbitals suitable for the geometry of covalent bonds. To find the hybridization of the central atom, we use the formula:
Where:
The value of H corresponds to the hybridization:
Central atom: Nitrogen (N). Valence electrons (V) = 5.
Structure: N is bonded to 3 O atoms. One bond is a double bond, but for hybridization count, we treat all bonds as single bonds. Monovalent atoms (M) = 0 (O is not monovalent).
Anionic charge (A) = 1.
Calculation:
H = 3 corresponds to sp² hybridization.
Central atom: Nitrogen (N). Valence electrons (V) = 5.
Structure: N is bonded to 2 O atoms. Monovalent atoms (M) = 0.
Cationic charge (C) = 1.
Calculation:
H = 2 corresponds to sp hybridization.
Central atom: Nitrogen (N). Valence electrons (V) = 5.
Structure: N is bonded to 4 H atoms. Monovalent atoms (M) = 4.
Cationic charge (C) = 1.
Calculation:
H = 4 corresponds to sp³ hybridization.
The hybridisation of orbitals of N atom in NO3–, NO2+ and NH4+ are sp², sp, sp³ respectively.
Therefore, the correct option is: sp², sp, sp³
Hybridization Formula:
Steric Number: Another way to determine hybridization is by calculating the steric number, which is the sum of the number of atoms bonded to the central atom and the number of lone pairs on it.
Key Theory: Hybridization explains the observed shapes of molecules by proposing that the valence atomic orbitals of the central atom mix to form new, degenerate (same energy) hybrid orbitals. These hybrid orbitals have defined directional properties that align with the molecular geometry predicted by VSEPR theory.