Understanding Isostructural Species
Isostructural species are molecules or ions that have the same molecular geometry or shape, even if their atoms or charges differ. To determine this, we analyze their electron pair geometry (based on VSEPR theory) and molecular structure.
Step 1: Analyze Each Pair
Pair 1: PCl4+ and SiCl4
- PCl4+: Central P has 4 bond pairs, no lone pairs. Tetrahedral geometry.
- SiCl4: Central Si has 4 bond pairs, no lone pairs. Tetrahedral geometry.
- Both are isostructural.
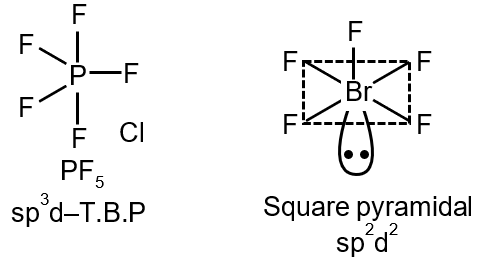
Pair 2: PF5 and BrF5
- PF5: Central P has 5 bond pairs, no lone pairs. Trigonal bipyramidal geometry.
- BrF5: Central Br has 5 bond pairs and 1 lone pair. Square pyramidal geometry.
- Different geometries → not isostructural.
Pair 3: CO32- and NO3-
- CO32-: Central C has 3 bond pairs, no lone pairs. Trigonal planar geometry.
- NO3-: Central N has 3 bond pairs, no lone pairs. Trigonal planar geometry.
- Both are isostructural.
Pair 4: AlF63- and SF6
- AlF63-: Central Al has 6 bond pairs, no lone pairs. Octahedral geometry.
- SF6: Central S has 6 bond pairs, no lone pairs. Octahedral geometry.
- Both are isostructural.
Step 2: Final Answer
The pair that is not isostructural is PF5 and BrF5.
Related Topics & Formulae
VSEPR Theory (Valence Shell Electron Pair Repulsion): Predicts molecular geometry based on electron pair repulsion. Key formula for steric number (SN):
Common geometries:
- SN=4, no lone pairs: Tetrahedral
- SN=5, no lone pairs: Trigonal bipyramidal
- SN=6, no lone pairs: Octahedral
- SN=5, 1 lone pair: Square pyramidal
- SN=4, 1 lone pair: Trigonal pyramidal
Isostructuralism: Depends on identical electron pair arrangement and molecular shape, regardless of atom type or charge.