The species having pyramidal shape is
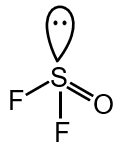
To identify which species has a pyramidal shape, we need to analyze their molecular geometry using VSEPR theory. Pyramidal geometry occurs when a molecule has 4 electron domains (3 bonding pairs + 1 lone pair) around the central atom.
Option 1: SiO32–
Central atom: Si (Group 14, 4 valence electrons)
O atoms contribute 0 electrons (each O forms double bond)
Charge: +2 electrons (from 2– charge)
Total valence electrons: 4 + 2 = 6
Electron domains: 3 (all bonding, no lone pairs)
Geometry: Trigonal planar
Option 2: OSF2
Central atom: S (Group 16, 6 valence electrons)
O contributes 0 electrons (double bond), F atoms contribute 1 each
Total valence electrons: 6 + 2 = 8
Electron domains: 3 bonding + 1 lone pair = 4 domains
Geometry: Pyramidal
Option 3: SO3
Central atom: S (Group 16, 6 valence electrons)
O atoms contribute 0 electrons (all form double bonds)
Total valence electrons: 6
Electron domains: 3 (all bonding)
Geometry: Trigonal planar
Option 4: BrF3
Central atom: Br (Group 17, 7 valence electrons)
F atoms contribute 1 each: 3 electrons
Total valence electrons: 7 + 3 = 10
Electron domains: 3 bonding + 2 lone pairs = 5 domains
Geometry: T-shaped
Only OSF2 has the required 3 bonding pairs + 1 lone pair configuration that gives pyramidal geometry.
The species with pyramidal shape is OSF2
VSEPR Theory: Valence Shell Electron Pair Repulsion theory predicts molecular geometry based on electron domain arrangement around central atoms.
Electron Domain Formula: