In allene (C3H4), the type(s) of hybridisation of the carbon atoms is (are)
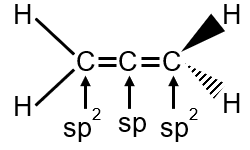
Allene has the structure H2C=C=CH2, with two double bonds between the three carbon atoms. The central carbon is bonded to two other carbons, and each terminal carbon is bonded to two hydrogen atoms.
Each terminal carbon (C1 and C3) is part of a double bond (one σ and one π bond) and bonded to two hydrogen atoms. The steric number (number of atoms bonded + lone pairs) is 3 (2 H atoms + 1 C atom), so hybridization is sp2.
Steric number calculation:
The central carbon (C2) is bonded to two other carbons via double bonds. Each double bond consists of one σ bond, so C2 has two σ bonds. The steric number is 2, indicating sp hybridization.
Steric number:
Terminal carbons (sp2) are trigonal planar with ~120° bond angles. The central carbon (sp) is linear with 180° bond angle between the two terminal carbons, consistent with allene's structure.
The hybridizations are sp and sp2 (central carbon is sp, terminal carbons are sp2).
Hybridization is determined by steric number (SN):
For SN=2: sp hybridization (linear geometry)
For SN=3: sp2 hybridization (trigonal planar geometry)
For SN=4: sp3 hybridization (tetrahedral geometry)
In allene, the central carbon has no lone pairs and two σ bonds (SN=2), while terminal carbons have three σ bonds each (SN=3).