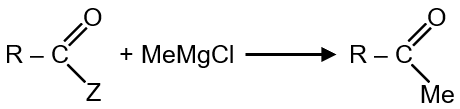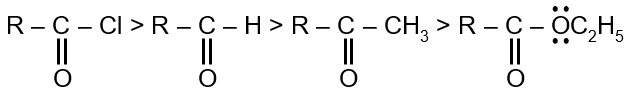Understanding Reaction Rate and Substituent Effects
This question involves determining which substituent Z makes the nucleophilic substitution reaction fastest. The reaction is:
This is a nucleophilic substitution reaction (likely SN2) where OH− attacks the carbon attached to Z.
Step 1: Identify the Reaction Mechanism
The nucleophile (OH−) is attacking a primary carbon (CH2−Z). Primary carbons typically undergo SN2 reactions. The rate of an SN2 reaction is highly dependent on the nature of the leaving group (Z).
Step 2: Understand the Role of the Leaving Group (Z)
In SN2 reactions, a better leaving group increases the reaction rate. A good leaving group is one that is stable and weak after it departs (i.e., it is the conjugate base of a strong acid).
The general rule is: The weaker the base, the better the leaving group.
Step 3: Analyze the Basicity of the Potential Leaving Groups
Let's compare the basicity of the conjugate bases of the options for Z.
- If Z = OC2H5, the leaving group is ethoxide ion (C2H5O−). It is a strong base and a very poor leaving group.
- If Z = H, the leaving group is hydride ion (H−). It is an extremely strong base and a very poor leaving group.
- If Z = CH3, the leaving group is methyl carbanion (CH3−). It is also an extremely strong base and a very poor leaving group.
- If Z = Cl, the leaving group is chloride ion (Cl−). Cl− is the conjugate base of a strong acid (HCl), making it a weak base and an excellent leaving group.
Step 4: Determine the Fastest Reaction
Since the SN2 rate is fastest with the best leaving group, and Cl− is by far the best leaving group among the options, the reaction will be fastest when Z is Cl.
Final Answer
The reaction is fastest when Z is Cl.
Related Topics
- Nucleophilic Substitution Reactions: Reactions where a nucleophile replaces a leaving group in a molecule. The two main mechanisms are SN1 (unimolecular) and SN2 (bimolecular).
- Leaving Group Ability: The capacity of an atom or group to depart with the bonding electrons. It is inversely related to the basicity of the group.
- Basicity: The tendency of a species to donate electrons or accept a proton. pKa of the conjugate acid is a common measure; lower pKa means stronger acid and weaker conjugate base (better leaving group).
Key Formulae and Theory
Relationship between Leaving Group Ability and Basicity:
A leaving group (LG) is good if its conjugate acid (H-LG) is a strong acid.
For example:
- HCl (pKa ≈ -7) is a strong acid → Cl− is a weak base and excellent LG.
- H2O (pKa ≈ 15.7) is a weak acid → HO− is a strong base and poor LG.
- Alkoxides (RO−) and carbanions (R−) are very strong bases and terrible leaving groups.
SN2 Rate Law:
The rate depends on the concentration of both the substrate and the nucleophile, as well as the nature of the leaving group and the substrate's steric hindrance.