Sodium ethoxide has reacted with enthanoyl chloride. The compound that is produced in the above reaction is :
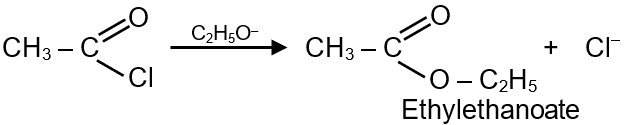
When sodium ethoxide (a strong nucleophile) reacts with ethanoyl chloride (an acyl chloride), it undergoes a nucleophilic substitution reaction. Ethanoyl chloride has the formula , and sodium ethoxide is . The ethoxide ion attacks the carbonyl carbon of ethanoyl chloride, leading to the substitution of the chloride ion and formation of an ester.
Step 1: Nucleophilic attack by ethoxide ion on the carbonyl carbon of ethanoyl chloride.
Step 2: The chloride ion leaves, and the product formed is ethyl ethanoate (an ester).
Final Answer: The compound produced is Ethyl ethanoate.
General reaction of acyl chloride with alcohol/alkoxide:
In this case, sodium ethoxide provides the ethoxide ion (), which acts as the nucleophile. Acyl chlorides are highly reactive due to the good leaving group ability of chloride ion and the electrophilic carbonyl carbon.