The major product H of the given reaction sequence is :
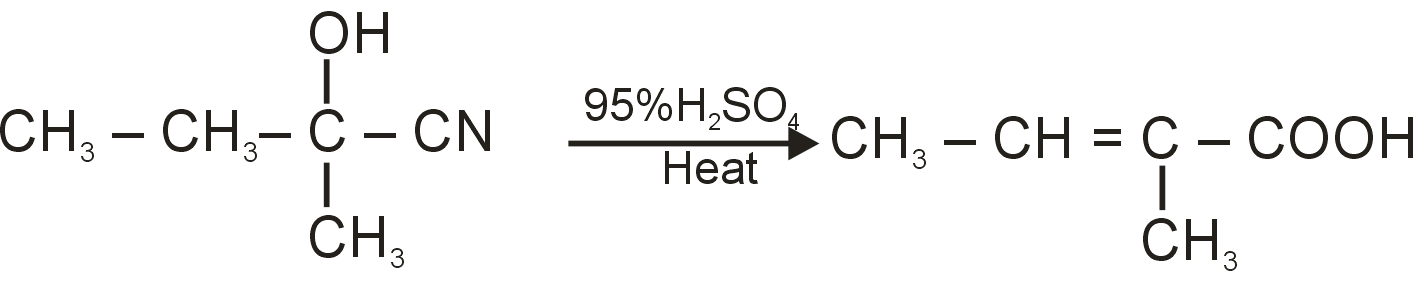
The given reaction sequence is:
This represents a ketone (Butan-2-one) undergoing a cyanohydrin reaction followed by hydrolysis with concentrated sulfuric acid.
Step 1: Cyanohydrin Formation (G)
The starting compound is Butan-2-one, a ketone: . It reacts with cyanide ion (). This is a nucleophilic addition reaction. The cyanide ion attacks the electrophilic carbonyl carbon, forming a cyanohydrin (G).
Reaction:
Intermediate G is: 2-Hydroxy-2-methylbutanenitrile
Step 2: Hydrolysis with Concentrated H₂SO₄ (H)
The cyanohydrin (G) is heated with 95% concentrated sulfuric acid. This is a classic condition for the hydrolysis of a cyanohydrin, which proceeds via a dehydration-hydrolysis mechanism. The hydroxyl group is protonated and lost as water, reforming a carbonyl group. The nitrile group () is then hydrolyzed to a carboxylic acid group ().
Overall Reaction:
Wait, that is incorrect. The final product is not the original ketone. The key point is that the carbon that was the carbonyl carbon becomes part of the new carboxylic acid. The original ketone had 4 carbons. The product is an acid with one fewer carbon. This is the Ritter-like hydrolysis of cyanohydrins, which yields carboxylic acids with the loss of one carbon atom (as CO₂ or formic acid).
The correct mechanism involves the dehydration to reform an unsaturated nitrile, followed by hydrolysis. The net result is that the molecule is cleaved, producing a carboxylic acid with one less carbon than the original ketone.
For a ketone R-COR', the cyanohydrin hydrolysis gives RCOOH + R'COOH. Since our ketone is CH₃CH₂COCH₃ (R = C₂H₅, R' = CH₃), the products should be Propanoic acid (CH₃CH₂COOH) and Acetic acid (CH₃COOH).
However, the question asks for the major product H. The reaction is intramolecular, and the major product is determined by the stability of the resulting carbocation during the dehydration step. The hydroxyl group is protonated and leaves, generating a carbocation at that carbon. The more stable carbocation will lead to the major product.
In our cyanohydrin, , the carbon bearing the OH is tertiary. Loss of water gives a stable tertiary carbocation: . This cation is attacked by the nitrile nitrogen, forming an nitrilium ion. Hydrolysis of this nitrilium ion gives the amide, which further hydrolyzes to the acid.
The major product H will be the acid derived from the more stable alkyl group. The tertiary carbocation can be represented as CH₃CH₂C⁺(CH₃)₂, which is more stable than any alternative. Hydrolysis gives the acid corresponding to that carbocation: (CH₃)₂CHCOOH (2-Methylpropanoic acid).
Let's trace the atoms: The original carbonyl carbon becomes the carboxylic acid carbon. For Butan-2-one, CH₃-CH₂-C(O)-CH₃, the carbonyl carbon is C3. After cyanohydrin formation and hydrolysis, this carbon is lost as CO₂? No, the mechanism shows the carbon from the CN group becomes the carboxylic acid carbon.
Correction: In the final hydrolysis, the nitrile carbon becomes the carboxyl carbon. The original ketone carbon becomes part of the alkyl group. Therefore, the major product H is a carboxylic acid where the alkyl group is derived from the more stable carbocation fragment.
Final Answer: The major product H is 2-Methylpropanoic acid .
Looking at the options, this corresponds to the structure: 
The major product H of the reaction sequence is 2-Methylpropanoic acid.

Key Concepts:
Important Theory: The hydrolysis of cyanohydrins with concentrated sulfuric acid is a specific reaction that leads to the degradation of the original carbonyl compound. It proceeds through the formation of a carbocation, followed by intramolecular capture by the nitrile group. The final carboxylic acid produced has its alkyl group determined by the stability of this carbocation, not simply by the structure of the original ketone.