The total number of alkenes possible by dehydrobromination of 3‑bromo-3-cyclopentylhexane using alcoholic KOH is :
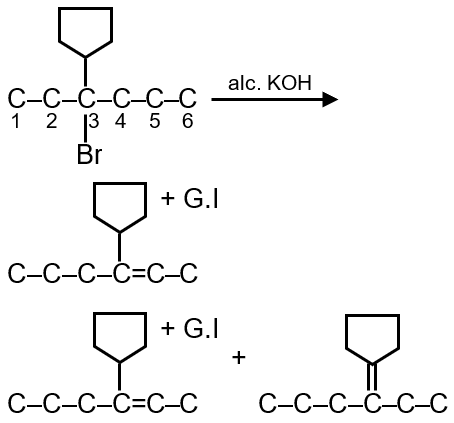
Dehydrobromination is an elimination reaction where a hydrogen halide (HBr) is removed from an alkyl halide using a base (like alcoholic KOH), forming an alkene. The possible alkenes depend on the number of distinct β-hydrogens available for elimination.
Step 1: Identify the structure. The compound is 3-bromo-3-cyclopentylhexane. The parent chain is hexane with a bromine and a cyclopentyl group both attached to carbon 3. So, carbon 3 is tertiary and chiral.
Step 2: Locate β-carbons. β-carbons are adjacent to the carbon bearing the bromine (C3). So, C2 and C4 are β-carbons.
Step 3: Count β-hydrogens. Each β-carbon has hydrogens that can be eliminated.
Step 4: Determine distinct elimination products.
So, total 3 alkenes are possible.
Step 5: Check for stereoisomers. The alkene formed with cyclopentyl might show E/Z isomerism? But in this case, the alkene is trisubstituted and the groups are different, so it can have E and Z isomers. However, the question asks for "number of alkenes", which typically includes stereoisomers if they are distinct.
For alkene between C3 and cyclopentyl carbon: one side has hexyl chain (C4-C5-C6), the other side has cyclopentyl. So, it is unsymmetrical and E/Z isomers are possible. Therefore, this elimination gives 2 alkenes (E and Z).
The other two alkenes (C2-C3 and C3-C4) are disubstituted and do not have stereoisomers.
So, total alkenes = 2 (from C2 and C4) + 2 (E and Z from cyclopentyl) = 4.
Final Answer: 4 alkenes are possible.
Elimination Reactions: These are reactions where a small molecule is removed from a substrate. Dehydrohalogenation is a common elimination reaction where HX is removed from an alkyl halide to form an alkene. The most common mechanism is E2, which is bimolecular and concerted.
Zaitsev's Rule: In elimination reactions, the more substituted alkene is generally the major product. However, the question is about the total number of possible alkenes, not the major one.
General reaction for dehydrohalogenation:
Number of alkenes depends on the number of unique β-hydrogens and the possibility of stereoisomers in the resulting alkene.