For given reaction sequence :
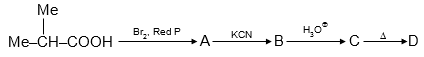
Correct option for this sequence :
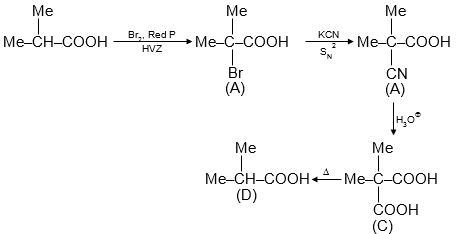
This reaction sequence involves the conversion of an alkene through several steps to form different carboxylic acid products. Let's analyze each step and identify the correct statements about the intermediates and products.
The starting compound is an alkene: . NBS is used for allylic bromination. The bromination occurs at the allylic position, which is the carbon adjacent to the double bond. The most stable allylic radical is formed, leading to bromination at the terminal carbon. So, product A is .
A undergoes nucleophilic substitution. The bromide is replaced by cyanide ion (CN⁻) to form a nitrile. So, product B is . This is not an α-bromo carboxylic acid; it is an unsaturated nitrile.
B is hydrolyzed under acidic conditions to convert the nitrile group to a carboxylic acid. So, product C is . This is an α,β-unsaturated carboxylic acid, not a vicinal dicarboxylic acid (which would have two COOH groups on adjacent carbons).
When C is heated, it undergoes decarboxylation. However, for α,β-unsaturated acids, heating can lead to isomerization or other reactions. In this case, upon heating, the double bond may shift, and decarboxylation occurs, leading to a saturated carboxylic acid. The major product D is or , which is isobutyric acid. This is indeed Me₂CH-COOH.
Allylic Bromination: NBS selectively brominates allylic positions via free radical mechanism. The stability of the radical follows: tertiary > secondary > primary.
Nucleophilic Substitution: Alkyl halides react with CN⁻ to form nitriles, which can be hydrolyzed to carboxylic acids: R-Br + CN⁻ → R-CN; R-CN + H₃O⁺ → R-COOH.
Decarboxylation: α,β-unsaturated acids upon heating may decarboxylate to form alkenes or, under certain conditions, yield saturated acids due to isomerization.