Trichloroacetaldehyde was subjected to Cannizzaro's reaction by using NaOH. The mixture of the products contains sodium trichloroacetate and another compound. The other compound is :
The cannizzaro product of given reaction yields 2, 2, 2-trichloroethanol.
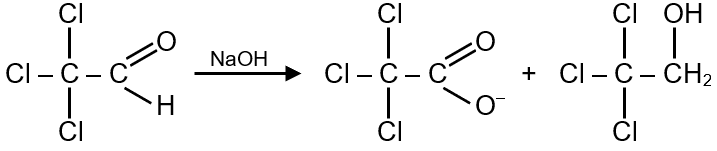
The Cannizzaro reaction is a chemical reaction that involves the disproportionation of an aldehyde lacking an alpha-hydrogen atom. In the presence of a strong base like NaOH, one molecule of the aldehyde is reduced to an alcohol, while another is oxidized to a carboxylic acid salt.
Trichloroacetaldehyde (also known as chloral) has the formula . It does not have an alpha-hydrogen because the carbon adjacent to the carbonyl group is bonded to three chlorine atoms, so it undergoes the Cannizzaro reaction.
Step 1: Write the balanced Cannizzaro reaction for trichloroacetaldehyde with NaOH.
Two molecules of trichloroacetaldehyde react with one molecule of NaOH. One aldehyde molecule is oxidized to sodium trichloroacetate, and the other is reduced to an alcohol.
Reaction:
Step 2: Identify the products.
The oxidation product is sodium trichloroacetate (), as given. The reduction product is the alcohol: trichloroethanol, which is named systematically as 2,2,2-trichloroethanol ().
Step 3: Compare with the options.
Final Answer: The other compound is 2,2,2-Trichloroethanol.
General Cannizzaro Reaction:
Where R is a group without alpha-hydrogen (e.g., aryl, CCl₃, etc.).
Trichloroacetaldehyde (Chloral) Structure:
Formula:
No alpha-hydrogen present, so undergoes Cannizzaro.
Product Identification:
Reduction product: (2,2,2-trichloroethanol)
Oxidation product: (sodium trichloroacetate)