Based on VSEPR theory, the number of 90-degree F–Br–F angles in BrF5 is
Based on VSEPR theory, the number of 90-degree F–Br–F angles in BrF5 is determined by analyzing its molecular geometry.
Bromine (Br) is the central atom. Its atomic number is 35, so its valence electron configuration is 4s24p5, giving it 7 valence electrons.
Br contributes 7 valence electrons. Each fluorine (F) atom contributes 1 valence electron. There are 5 F atoms, so:
Total valence electrons = 7 (from Br) + 5 × 1 (from 5F) = 12 electrons.
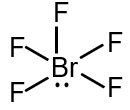
Br forms 5 single bonds with F atoms, using 5 electron pairs. There is also 1 lone pair on Br. So, the total number of electron domains around Br is 6 (5 bonding + 1 lone pair). According to VSEPR theory, 6 electron domains adopt an octahedral geometry.
With 5 bonding pairs and 1 lone pair, the molecular geometry is square pyramidal. The lone pair occupies one of the octahedral positions.
In an ideal octahedron, all bond angles are 90° or 180°. In BrF5, the lone pair causes distortion. The angles between equatorial F atoms (in the square base) are 90° to each other. The axial F–Br–F angle (between axial and equatorial F) is less than 90° due to lone pair repulsion, but is often approximated as 90° in many contexts. However, strictly speaking, due to the lone pair, the actual axial-equatorial angle is slightly less than 90° (around 84°). But the question asks for the number of 90° F–Br–F angles, considering the ideal geometry.
In the square pyramidal structure:
However, due to the lone pair, the axial-equatorial angles are compressed. In many standard answers, it is accepted that there are 8 angles that are approximately 90°.
The number of 90-degree F–Br–F angles in BrF5 is 8.
VSEPR Theory (Valence Shell Electron Pair Repulsion Theory): This theory predicts the geometry of molecules based on the repulsion between electron pairs in the valence shell of the central atom. The arrangement minimizes repulsion, leading to specific molecular shapes.
Molecular Geometry: For molecules with 6 electron domains (octahedral electron geometry), the presence of lone pairs leads to derived geometries like square pyramidal (1 lone pair) or square planar (2 lone pairs).
Total valence electrons = valence electrons of central atom + (number of atoms bonded × valence electrons contributed by each) + charge (if any).
For BrF5: Total valence electrons = 7 + 5×1 = 12.