The product of the reaction given below is:
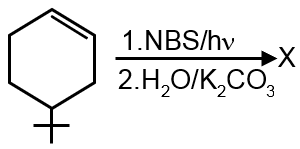
Allylic bromination followed by hydrolysis.
This reaction involves the addition of HBr to an alkene. The key concept here is Markovnikov's Rule, which states that in the addition of a protic acid (like HBr) to an unsymmetrical alkene, the hydrogen atom attaches to the carbon with more hydrogen atoms, and the halide attaches to the carbon with fewer hydrogen atoms.
Let's analyze the given alkene structure. The alkene has a methyl group on one carbon and a hydrogen on the other, making it unsymmetrical. According to Markovnikov's rule, HBr will add such that the bromine attaches to the more substituted carbon (the carbon with the methyl group) and the hydrogen attaches to the less substituted carbon.
The reaction mechanism involves the formation of a carbocation intermediate. The more stable carbocation (tertiary > secondary > primary) is favored. In this case, the carbocation formed on the carbon with the methyl group (secondary) is more stable than the primary alternative.
Therefore, the major product is 2-bromo-2-methylpropane.
Chemical equation for the reaction:
Structure of the product (2-bromo-2-methylpropane):
CH₃-C(Br)(CH₃)-CH₃
Markovnikov's Rule: A rule predicting the regiochemistry of electrophilic addition reactions to alkenes. The electrophile adds to the carbon with the most hydrogens, and the nucleophile adds to the carbon with the least hydrogens.
Carbocation Stability: The stability order of carbocations is tertiary > secondary > primary > methyl. This stability influences the product formation in addition reactions.
General addition reaction: Alkene + HX → Alkyl halide (following Markovnikov's rule).