The major product of the following reaction is:
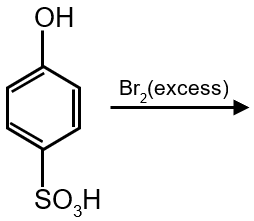
–SO3H will be replaced by Br this is called ipso effect.
This reaction involves an alkene undergoing electrophilic addition with HBr. The alkene is unsymmetrical (1-methylcyclopentene), so we must consider Markovnikov's rule to predict the major product.
Step 1: Formation of the Electrophile and Carbocation
The π electrons of the alkene attack the electrophilic hydrogen of HBr. This results in the formation of a carbocation intermediate. The stability of this carbocation determines the major product.
The alkene is 1-methylcyclopentene. The more substituted carbon (tertiary) will form the more stable carbocation.
Step 2: Nucleophilic Attack
The bromide ion (Br⁻) then acts as a nucleophile and attacks the stable tertiary carbocation, forming the final alkyl bromide product.
Final Answer: The major product is 1-bromo-1-methylcyclopentane. This corresponds to the structure where the bromine is attached to the tertiary carbon (the same carbon as the methyl group). This is the third option in the list of possible products.
Markovnikov's Rule:
In the electrophilic addition of HX to an unsymmetrical alkene, the hydrogen atom adds to the carbon of the double bond that has the greater number of hydrogen atoms, and the halogen adds to the carbon with fewer hydrogen atoms. The underlying reason is the formation of the more stable carbocation intermediate.
Carbocation Stability:
The order of stability of carbocations is a fundamental concept that dictates the regiochemistry of such reactions. Stability increases with the number of alkyl groups attached to the positively charged carbon due to hyperconjugation and the inductive effect.
(Tertiary > Secondary > Primary > Methyl)
The general reaction for electrophilic addition is: