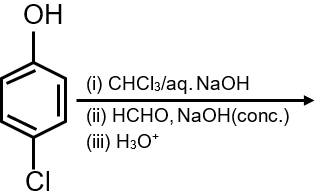
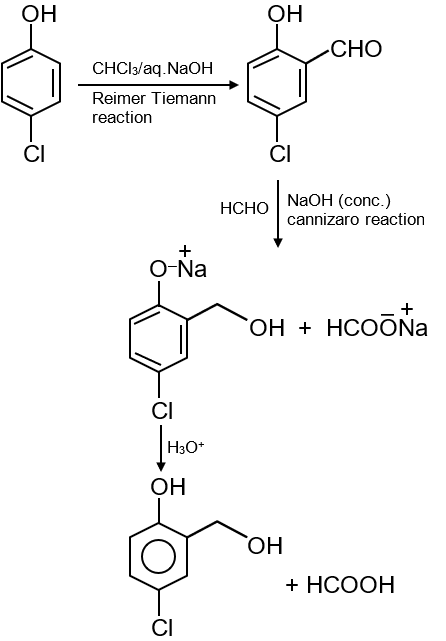
The major products of the following reaction are :
The given reaction involves a conjugated diene (1,3-butadiene derivative) undergoing electrophilic addition with HBr. This follows the mechanism of 1,2-addition and 1,4-addition due to resonance stabilization of the allylic carbocation intermediate.
Step 1: Protonation of the diene by HBr to form a resonance-stabilized allylic carbocation.
The initial protonation occurs at one of the terminal carbons, generating a carbocation that is resonance-stabilized:
Step 2: Nucleophilic attack by bromide ion (Br⁻) on the carbocation.
Br⁻ can attack either carbon of the resonance hybrid, leading to two products:
Final Answer: The major products are the 1,2-addition and 1,4-addition products. For the given diene (isoprene/2-methyl-1,3-butadiene), the 1,4-adduct is thermodynamically more stable and predominates under equilibrium conditions. The correct option is the third one, showing both possible adducts with emphasis on the conjugated product.
General Reaction: Conjugated diene + HX → 1,2-adduct + 1,4-adduct
Resonance Energy: Allylic carbocations are stabilized by resonance, lowering the activation energy.
Product Ratio: Depends on temperature (low temp favors 1,2; high temp favors 1,4) and stability of products.
Markovnikov's Rule: The electrophile (H⁺) adds to the less substituted end of the diene, forming the more stable carbocation.