What will be the major product in the following mononitration reaction?
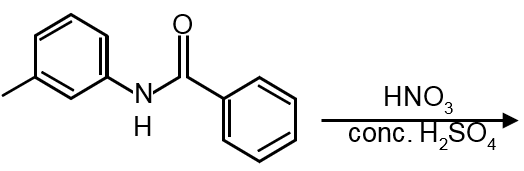
‘EAS’ first ring is activated and second is deactivated attack at para position of activated ring.
The given reactant is a disubstituted benzene ring with two substituents: a methoxy group (-OCH3) and a nitro group (-NO2). We need to determine the position where the new nitro group will attach during mononitration.
Substituents on benzene influence electrophilic substitution (like nitration) through two effects:
Since the methoxy group is a much stronger director than the nitro group, it will dominate the orientation of the incoming electrophile (NO2+).
The two substituents are meta to each other. The methoxy group will direct the new nitro group to its ortho and para positions. We must check which of these positions is sterically and electronically favored.
The possible positions for attack relative to the -OCH3 group are labeled below (the nitro group is fixed at the meta position):
Ortho to -OCH3: This position is also meta to the existing -NO2 group. A meta-director (like -NO2) weakly deactivates the meta position.
Para to -OCH3: This position is ortho to the existing -NO2 group. This is highly unfavorable due to the strong electron-withdrawing effect and significant steric hindrance from the nitro group.
Therefore, the ortho position to -OCH3 (which is meta to -NO2) is the major product. It is sterically less crowded and electronically more favorable than the para position.
The major product is the one where the new nitro group is attached ortho to the methoxy group and meta to the existing nitro group. This corresponds to the structure in the second option.
The general mechanism for nitration involves the attack of the electrophile NO2+ (nitronium ion) on the aromatic ring.
Activating (Ortho/Para Directors): -OH, -OR, -NH2, -alkyl groups. They donate electrons to the ring, making it more reactive.
Deactivating (Meta Directors): -NO2, -CN, -COOH, -COR, -SO3H. They withdraw electrons from the ring, making it less reactive.
Exception: Halogens (-Cl, -Br) are deactivating but ortho/para directors due to their resonance effect.