The major product of the following reaction is :
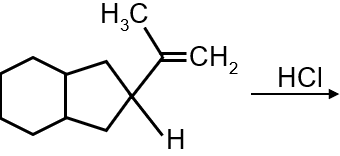
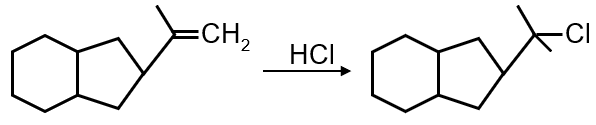
The given reaction involves a conjugated diene (1,3-butadiene derivative) undergoing electrophilic addition with HBr. The key concept is 1,2-addition vs 1,4-addition under kinetic vs thermodynamic control.
Step 1: Protonation to form allylic carbocation
HBr adds a proton (H⁺) to one of the terminal carbons of the diene, forming a resonance-stabilized allylic carbocation.
Step 2: Nucleophilic attack by bromide ion
Br⁻ attacks the carbocation. At low temperature (kinetic control), the reaction is faster and gives the 1,2-addition product. At higher temperature (thermodynamic control), the more stable 1,4-addition product dominates.
Since no temperature is specified, we assume standard conditions where the 1,4-adduct is the major product due to its greater stability (more substituted double bond).
Final Answer: The major product is the 1,4-addition product: 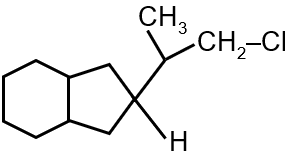
General reaction: Conjugated diene + HX → 1,2-adduct + 1,4-adduct
Kinetic product: Forms faster, favored at lower temperatures
Thermodynamic product: More stable, favored at higher temperatures
Stability order: Allylic carbocation > primary carbocation due to resonance