The magnetic moment (spin only) of [NiCl4]2– is :
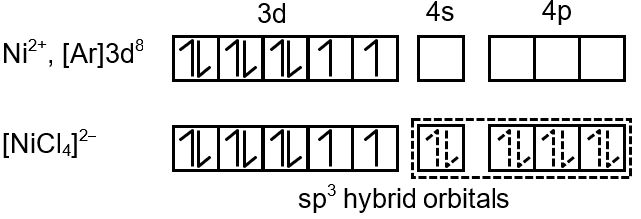
In the paramagnetic and tetrahedral complex [NiCl4]2–, the nickel is in +2 oxidation state and the ion has the electronic configuration 3d8. The hybridisation scheme is as shown in figure.
The magnetic moment (spin only) is calculated using the formula: BM, where is the number of unpaired electrons. For coordination compounds, we first determine the oxidation state of the metal, its electron configuration, and the geometry (which affects splitting of d-orbitals) to find unpaired electrons.
Step 1: Find oxidation state of Nickel (Ni)
Let oxidation state of Ni be . Chlorine (Cl) has -1 charge. The complex has overall charge -2.
Equation:
Solving: →
So, Ni is in +2 oxidation state.
Step 2: Write electron configuration of Ni2+
Atomic number of Ni is 28. Electronic configuration: [Ar] 3d8 4s2.
For Ni2+, two electrons are removed from 4s orbital first. So, configuration: [Ar] 3d8.
Step 3: Determine geometry and ligand field effect
Cl– is a weak field ligand. [NiCl4]2– has tetrahedral geometry (common for Ni2+ with Cl–).
In tetrahedral field, splitting is small (weak field), so electrons remain unpaired (high spin). For d8 in tetrahedral geometry, there are 2 unpaired electrons.
Explanation: The d orbitals split into e and t2 in tetrahedral field. With 8 electrons, filling is: e4 t24. Since t2 orbitals can hold 6 electrons, with 4 electrons, they are arranged with 2 unpaired (Hund's rule).
Step 4: Calculate number of unpaired electrons ()
From above, .
Step 5: Compute magnetic moment using spin-only formula
BM (approximately 2.82 BM).
Final Answer: 2.82 BM
Spin-only magnetic moment formula: BM, where is number of unpaired electrons.
For tetrahedral complexes: Splitting is smaller compared to octahedral. Weak field ligands often result in high spin complexes.
For Ni2+ (d8): In tetrahedral geometry, it has 2 unpaired electrons. In octahedral geometry with weak field ligands, it also has 2 unpaired electrons, but with strong field ligands (e.g., CN–), it has 0 unpaired electrons.
Common weak field ligands: Cl–, Br–, I–, H2O. Strong field ligands: CN–, CO, NH3.