Concept Explanation: Identifying Paramagnetic Compounds
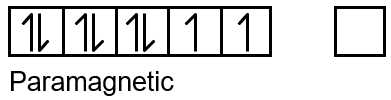
Paramagnetic compounds have unpaired electrons and are attracted to a magnetic field. To determine if a compound is paramagnetic, we analyze the electron configuration of the central metal ion, considering its oxidation state and the geometry imposed by the ligands (which affects the splitting of d-orbitals in crystal field theory).
Step-by-Step Analysis:
Step 1: List the compounds and identify the central metal ion and its oxidation state.
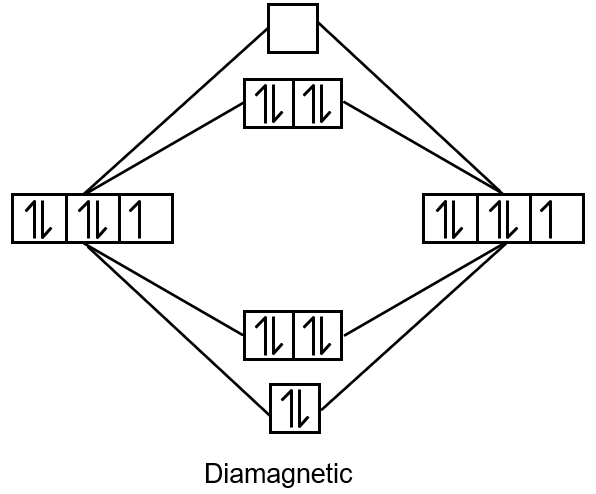
- [Ni(CO)4]: Nickel carbonyl. Ni is in 0 oxidation state. Electron configuration of Ni(0) is [Ar] 3d8 4s2. With strong field ligand CO, it forms tetrahedral complex. In tetrahedral field, d8 has two unpaired electrons? Actually, for Ni(0) d10? Wait, Ni atomic number 28: Ni0 has 28 electrons. Configuration: 1s22s22p63s23p63d84s2? But in coordination compounds, the 4s electrons are lost before 3d? For Ni(0), it is d10? Let's calculate: Ni atom has 28 electrons. In Ni(CO)4, Ni is in zero oxidation state, so it has 28 electrons. The configuration is [Ar] 3d8 4s2? But CO is a strong field ligand, and it causes pairing. Actually, Ni(CO)4 is diamagnetic because it has tetrahedral geometry and d10 configuration? How? Ni(0) must have 28 electrons. The common configuration for Ni(0) is 3d10? Yes, because in atoms, the 4s orbital is filled before 3d, but for Ni0, the configuration is [Ar] 3d8 4s2? That would be paramagnetic. However, in Ni(CO)4, the Ni is considered to have configuration 3d10 due to back bonding? Actually, it is known to be diamagnetic, so it has no unpaired electrons. So, it is not paramagnetic.
- [NiCl4]2-: Tetrachloronickelate(II). Ni2+ has d8 configuration. Cl- is weak field ligand. Tetrahedral geometry. For d8 in tetrahedral field, high spin, two unpaired electrons. Paramagnetic.
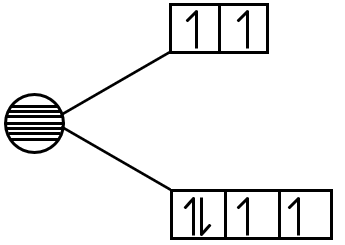
- [Co(NH3)4Cl2]Cl: Here, Co is in +3 oxidation state (since NH3 is neutral, Cl is -1, so to balance charge, Co is +3). Co3+ has d6 configuration. NH3 is strong field ligand. Octahedral geometry. Low spin d6 has no unpaired electrons. Diamagnetic.
- Na3[CoF6]: Hexafluorocobaltate(III). Co3+ d6. F- is weak field ligand. Octahedral geometry. High spin d6 has four unpaired electrons. Paramagnetic.
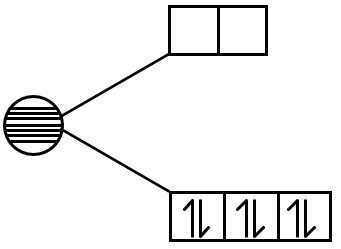
- Na2O2: Sodium peroxide. Contains O22- ion. This ion has all electrons paired (bond order 1). Diamagnetic.
- CsO2: Cesium superoxide. Contains O2- ion. This ion has one unpaired electron (bond order 1.5). Paramagnetic.
Step 2: Count the paramagnetic compounds.
From above: [Ni(CO)4] is diamagnetic. [NiCl4]2- is paramagnetic. [Co(NH3)4Cl2]Cl is diamagnetic. Na3[CoF6] is paramagnetic. Na2O2 is diamagnetic. CsO2 is paramagnetic.
So, paramagnetic are: [NiCl4]2-, Na3[CoF6], CsO2. That is 3 compounds.
Final Answer: The total number of paramagnetic compounds is .
Related Topics:
- Crystal Field Theory: Explains how ligands affect the d-orbitals of metal ions, leading to splitting and electron pairing.
- Oxidation States: Determining the charge on the central metal ion is crucial for finding its d-electron count.
- Magnetic Properties: Diamagnetic (all electrons paired) vs. Paramagnetic (unpaired electrons present).
- Coordination Geometry: Tetrahedral vs. Octahedral complexes have different splitting patterns.
- Superoxide and Peroxide Ions: O2- has an unpaired electron, while O22- does not.
Key Formulae and Theory:
For a metal ion in a complex:
- Number of d electrons = Atomic number - oxidation state (for first row transition metals, subtract from 21 to 30? Actually, for Ni, atomic number 28, Ni2+ has 26 electrons, so d electrons = 8).
- In tetrahedral field, splitting is small, so often high spin.
- In octahedral field, with strong field ligands (like CO, NH3), pairing occurs (low spin). With weak field ligands (like Cl-, F-), high spin.
- For dn configurations:
- d6 octahedral low spin: paired (diamagnetic).
- d6 octahedral high spin: 4 unpaired (paramagnetic).
- d8 tetrahedral: always two unpaired (paramagnetic).
- d10: all paired (diamagnetic).
- For O2- superoxide: bond order = 1.5, unpaired electron.
- For O22- peroxide: bond order = 1, all paired.